Mega Malaria Extravaganza
aka Tropical Travel Trouble 007
When you think tropical medicine, malaria has to be near the top. It can be fairly complex and fortunately treatment has become a lot simpler. This post is designed to walk you through the basic principals with links to more in depth teaching if your niche is travel medicine, laboratory diagnostics or management of severe or cerebral malaria.
If you stumbled on this post while drinking a cup of tea or sitting on the throne and want a few basic facts then skip to the end for a brief summary of the salient teaching points. As always do not hesitate to contact a specialist but hopefully armed with a little bit more knowledge.
Q1. What is malaria and can you name the five types that infect humans?
Answer and interpretation
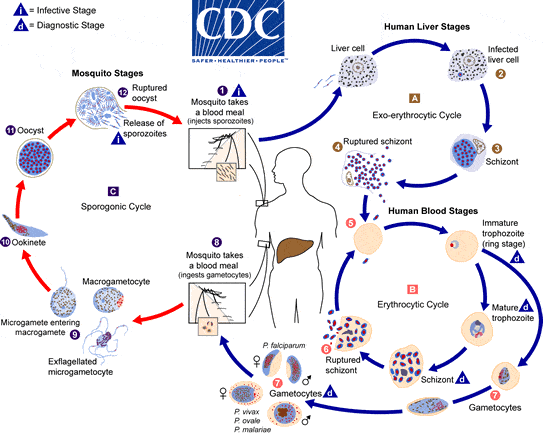
Malaria is a protozoan parasite transmitted by the female anopheles mosquito. Like many vectors in tropical medicine it is the female who is responsible, they require blood meals in order to produce their eggs. Males feed exclusively on fruit.
There are 5 types known to infect humans:
- Plasmodium falciparum (most dangerous form and associated with death).
- Plasmodium vivax (causes relapsing fevers due to hypnozoites that hide in the liver).
- Plasmodium ovale (causes relapsing fevers due to hypnozoites that hide in the liver).
- Plasmodium malariae and Plasmodium knowlesi appear identical under microscopy and require PCR to differentiate them.
Q2. What is the life cycle of malaria?
Q3. Where are the different types of malaria, and why is this important to know for my patient?
Answer and interpretation
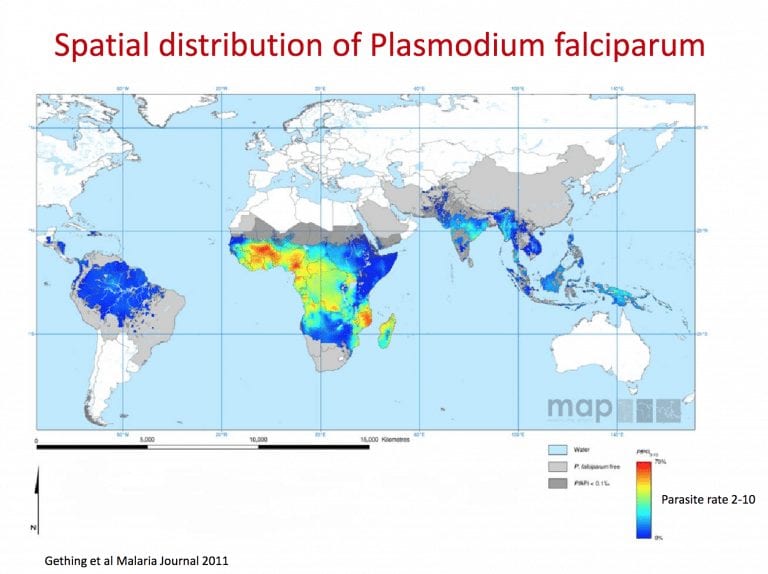
- Falciparum: throughout tropics, sub-Saharan Africa, SE Asia and S.America.
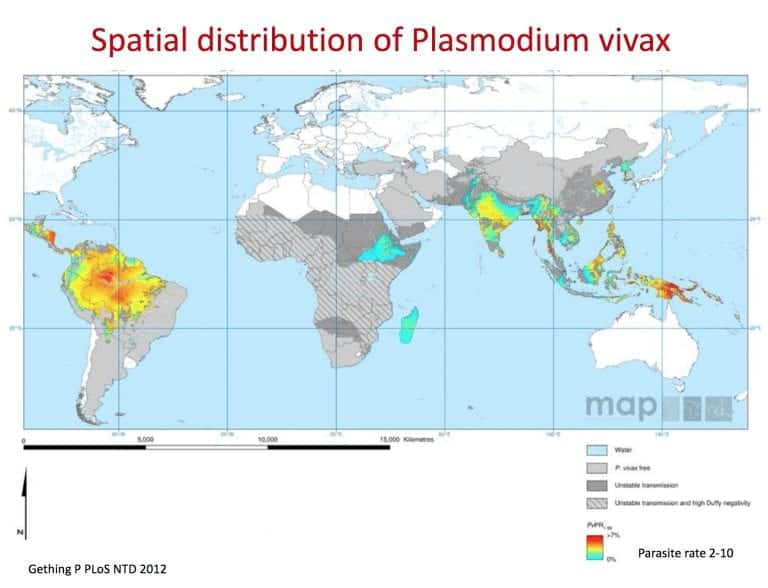
- Vivax: Central/S America, African horn, India/Pakistan, SE Asia.
- Ovale: Only in West Africa.
- Malariae: Worldwide.
- Knowlesi: Borneo and peninsular Malaysia.
It is important to know whether your patient is at risk of P.Falciparum, which is the most significant for causing cerebral malaria and mortality. Or, is your patient having relapsing fevers a year after being in West Africa or India, then you’ll need to consider hypnozoites in the liver from P. ovale or P. vivax.
Q4. What are the signs and symptoms of malaria?
Answer and interpretation
Uncomplicated:
- Fever in 90%.
- Classically described as a regular periodic fever, with tertian fever (P.falciparum, P.vivax and P.ovale) causing fevers every third day (every 48 hours) and quartan malaria (P.malariae) with fever every 4th day (every 72 hours). This periodicity only occurs if patients present late; it is unreliable in most instances.
- Non-specific flu-like symptoms: Headache, rigors, myalgia, malaise, minor gastrointestinal symptoms and jaundice from haemolysis. Splenomegaly and signs of anaemia may be seen clinically.
- Requires a high index of suspicion with some cases of patients returning to the UK having been diagnosed a year after returning from an endemic country.
- In endemic areas it has become common to say ‘I have malaria‘ when you have a fever.
Severe:
- Mortality of severe malaria approaches 100% and can occur within hours. With prompt treatment and supportive care this falls to 10-20% overall.
- Often thought of in P. falciparum although there have been cases in P. vivax and P. knowlesi. P. falciparum is particularly troublesome because the infected RBCs are prevented from entering the spleen by ‘sequestration’ – deformities in the red cell membrane which results in adhesions to capillary walls resulting in hypoxia to downstream tissues and potentially the relapse of increased cytokines.
Severe P. falciparum is defined as:
- GCS <11 in adults or a Blantyre coma score <3 in children.
- Cerebral malaria is misdiagnosed 25% of the time based on autopsies in Malawi. A good clue is to perform a fundoscopy looking for malarial retinopathy (white centered hemorrhages, vessel changes and whitening).
- Generalised weakness, the person can not walk without assistance.
- More than 2 convulsions in 24 hours.
- Acidosis: base deficit >8 mEq/L of bicarbonate <15 mmol/L or lactate >5 mmol/L.
- Hypoglycaemia <2.2 mmol/L (<40 mg/dL).
- Severe malarial anaemia: haemoglobin concentration <5 g/dL or a haematocrit of <15% in children <12 years of age (<7 g/dL and <20% , respectively, in adults) with a parasite count >10,000/uL.
- Raised creatinine >265 umol/L (3 mg/dL) or urea >20 mmol/L.
- Raised bilirubin >50 umol/L (3 mg/dL) with a parasite count >100,000/uL.
- Pulmonary oedema either radiographically confirmed or saturations <92% on room air with a respiratory rate >30/min.
- Significant bleeding.
- Shock: capillary refill >3 seconds. Evidence of poor perfusion to their peripheries or s systolic blood pressure <70 mmHg in children or <80 mmHg in adults.
- Hyperparasitaemia: >2%.
Severe Vivax: Same as falciparum but no parasite density threshold.
Severe Knowlesi:
- Parasite density >100,000/uL.
- Jaundice and parasite density > 20,000/uL.
Q5. What is the mechanism of “immunity” to malaria?
Answer and interpretation
Innate immunity:
- Haemoglobinopathies (Thalessaemia).
- RBC enzyme deficiencies (G6PD).
- RBC surface components (Duffy blood group).
- Sickle cell haemoglobin.
Incomplete immunity from prior repeated exposure:
- In populations who are continually exposed to malaria (inoculation rate >10/year) a partial immunity to clinical disease and a reduced risk of developing severe malaria can occur. Clinically you will see disease in young children but adolescents and adults seldom suffer clinical disease. This immunity is lost if the person moves out of an endemic area.
- In pregnancy the placental tissue has a preference from PfEMP1. Once antibodies are developed they are usually protected from malaria in pregnancy but until this stage there is higher morbidity and mortality for both the mother and foetus.
Q6. Why does pregnancy increase susceptibility to malaria?
Answer and interpretation
- Physiological changes – increased ‘host signature’ helps mosquitoes to detect the host (increased production of oestrogen and cortisol)
- Behaviour changes – leave protection of bed nets during the night twice as frequently to urinate.
- Pregnancy causes a generalised immuno-suppression (reduced type 1 helper cells)
- Acquisition of protective immunity against placental malaria is only acquired over successive pregnancies.
- Parasites accumulate and replicate in the placenta, higher densities and mature stages. Interferes with oxygen and nutrient transfer. As the parasite sequesters in the placenta the sensitivity of our diagnostic methods falls dramatically. A high index of suspicion and multiple blood films are required.
Malaria is more severe in pregnancy and requires prompt treatment. Awaiting WHO policy update but current evidence states risk-benefit balance favours ACTs in all 3 trimesters for proven malaria. When compared to quinine, artemisinin is not associated with an increased risk of spontaneous abortions, or still births or congenital malformations.
Intermittent Preventative Treatment in pregnancy (IPTp or SP-ITPp) if offered to all women in endemic areas of Africa in the first and second pregnancy. The reason ‘screen and treat’ is not adopted is due to the reduced sensitivity of our diagnostic tests, sometimes down to 50% in certain populations.
- Sulfadoxine-pyrimathamine (SP), monthly from the second trimester.
- HIV+ve women receive daily cotrimoxazole instead of IPTp.
- IPTp reduces maternal malaria by 10%, placental malaria by 52%, LBW by 29% and increases mean birth weight by up to 79g.
- Given every scheduled visit in the 2nd and 3rd trimester.
- Folic acid reduced to 0.4mg/day when using SP.
It has also been proposed to use a similar strategy for infants <12 months to receive SP in conjunction with their vaccinations of DTP and measles in moderate to high transmission areas. This is called Intermittent Preventative Treatment for infants (SP-IPTi). Do not give to infants who are already on co-trimoxazole.
Some children under the age of 6 years in seasonal malaria will receive seasonal malaria chemoprevention (SMC) with monthly amodiaqunie + SP during the transmission season. Do not give to infants who are already on co-trimoxazole.
Q7. What is Blackwater Fever?
Answer and interpretation
Blackwater fever (BWF) is a severe clinical syndrome, characterised by intravascular hemolysis, hemoglobinuria, and acute renal failure. Haemoglobin is eliminated via the kidneys causing a black urine – the Blackwater Fever.
It was a particular problem in European expatriates chronically exposed to Plasmodium falciparum and drinking tonic water which contained quinine. Once given treatment dose of quinine for malaria a number of expatriate developed BWF and several theories were hypothesised. BWF virtually disappeared after 1950, when chloroquine superseded quinine.
Q8. How do you diagnose malaria?
Answer and interpretation
There are two methods routinely used, light microscopy (thick film and thin films) and immunochromatographic rapid diagnostic tests (RDTs). The latter detects parasite-specific antigens or enzymes that are either genus or species specific.
Thick and thin is the gold standard and any competent microscopist should see malaria parasites.
RDT target antigens:
- Some will detect Histidine-rich-protein-2 (HRP-2) which is only expressed by plasmodium falciparum.
- Parasite lactate dehydrogenase (pLDH) can be P.falciparum specific (Pf-pLDH) or P.vivax specific pLDH (Pv-pLDH)
- Pan-specific pLDH will detect all malaria species.
RDTs are useful when you don’t have a microscopist, a malaria parasite is seen but you are unsure of the species or if a patient has had incomplete antimalarial treatment.With incomplete treatment the thick and thin films may be negative and repeat films should be performed if suspicion is high (every 6-12 hours) or an RDT that can detect LDH is the first to turn negative, indicating potential early treatment benefit. PfHRP2 will be positive for a couple of weeks after treatment, but again could indicate treatment success if negative after this time period.
If both a slide examination and an RDT is negative it is extremely unlikely the patient has malaria and other causes should be sought.Be aware the traditional teaching was for 3 thick and thin films 12 hours apart and your institution may still practise this strategy.
PCR is available but is largely used in research or looking at drug resistance.• If you are out in the field and need some microscopy tips see the WHO learners guide, also test yourself with their malaria quiz.
Q9. How do you treat malaria?
Answer and interpretation
Uncomplicated:
If you want to only remember one drug – oral Artesunate combination therapy (ACT) on day 1,2,3 for children and adults except women in the first trimester (simple, although reports of resistance in SE Asia to artesunate are emerging and longer courses are required or new ACT regimens that are not yet licensed – see your local guidelines).
- Artemether + lumefantrine.
- Artesunate + amodiaquine.
- Artesunate + mefloquine.
- Dihydroartemisinin + piperaquine (in children <25kg this should be dosed as 2.5mg/kg of dihydroartemisinin and 20mg/kg of piperaquine). Can prolong the QT interval.
- Artesunate + sulfadoxine-pyrimathamine (SP). Women taking 5mg of folic acid greatly reduce the efficacy of SP and this combination should be avoided. 0.4mg of folic acid is okay.
These five ACT regimens are used because artemisinin rapidly clears the blood of parasites by a factor of 10,000 each 48hr asexual cycle. It is also active against the sexual stages of the parasite that meditate onward transmission to mosquitoes. The longer-acting partner drug clears the remaining parasites and provides protection against artemisinin resistance.
- In low transmission countries give a single dose of Primaquine 0.25mg/kg in addition to ACT for anti-gametocyte effect as part of malaria elimination programme for P.falciparum. (G6PD testing is not required but currently not approved for pregnant and breast feeding women and children <6 months).
- Chloroquine can be used to treat P. vivax, P. ovale, P. malariae and P. knowlesi if sensitive but to keep things simple you can just use ACT (except women in the first trimester). Chloroquine is dosed at 10mg/kg on day 1, day 2 and 5mg/kg on day 3.
- To prevent a relapse of P. vivax or P. ovale use a 14 day course of primaquine 0.25-0.5mg/kg unless G6PD is a possibility (i.e. infants, pregnant women, women breast feeding and those with confirmed G6PD). If G6PD is a possibility or confirmed treat with primaquine but at 0.75mg/kg once a week for 8 weeks with close medical supervision. It has dose related complications.
- Anyone with P. falciparum should be admitted for 24 hours due to the risk of sudden deterioration at the beginning of treatment.
Special Populations:
- Pregnancy and breast feeding
- In the 1st trimester of pregnancy give quinine +/- clindamycin 10mg/kg BD (P.falciparum) for 7 days (guidelines may change later in 2018 to ACT as new safety data is about to emerge).
- To eradicate P. ovale and P. vivax in pregnant or breast feeding women, consider weekly chemoprophylaxis with chloroquine until delivery and breastfeeding is complete, on the basis of G6PD status treat with primaquine to prevent a future relapse.
- Children < 5kg get dosed at 5kg for ACT.
- HIV patients, avoid artesunate + SP if on co-trimoxazole. Or artesunate + amodiaquine if they are also on efavirenz or zidovudine. HIV patient drug interactions.
Severe Malaria:
Intravenous or intramuscular artesunate for at least 24 hours until oral medication can be tolerated (usually a complete 3 day course of ACT on top of the parenteral treatment and add primaquine in areas of low transmission).
Artesunate slow IV injection (3 to 5 minutes) or, if not possible, slow IM injection, into the anterior thigh.
- Children less than 20 kg: 3 mg/kg/dose.
- Children 20 kg and over and adults: 2.4 mg/kg/dose.
- One dose on admission (H0).
- One dose 12 hours after admission (H12).
- One dose 24 hours after admission (H24).
- Then one dose once daily.
- Administer at least 3 doses, then, if the patient can tolerate oral route, change to an ACT.
- If artesunate is not available use artemether over quinine. All of these drugs can be given intramuscular if you are in a remote area (IM is not appropriate in shock). Artemether is 3.2 mg/kg intramuscular loading and then 1.6 mg/kg intramuscular daily thereafter. Once the patient can tolerate oral medications change to ACT.
- If in the community and IM artesunate or artemether is not available, children under the age of 6 years can have a single rectal dose at 10mg/kg of artesunate while awaiting transfer (reduced mortality by 25%). In older children and adults it was associated with more deaths.
- Children 3 to 6 kg = 1 suppository of 50mg.
- Children 6 to 11 kg = 2 suppositories 50mg.
- Children 11 to 20 kg = 1 suppository 200mg.
Other options include the cinchona alkaloids (quinine and quinidine) but the largest clinical trials have shown substantial reduction in mortality with artesunate when compared with quinine.
- Quinine loading dose is 20 mg salt/kg BD (do not load if the patient has received oral quinine or mefloquine in the previous 24 hours), followed by 10 mg salt/kg Q8hrly as maintenance, starting 8 hours from the first dose. If there is no improvement in the patient’s condition within 48 hours the dose should be reduced by one third e.g. 10mg salt/kg Q12 hours.
- Rapid infusion of quinine is toxic (hypotension, blindness and deafness) and is usually administered in 5% dextrose over 4 hours making sure the rate does not exceed 5 mg salt/kg per hour. Hence the intramuscular route can be quite attractive in resource-limited settings.
- Quinine also causes hypoglycaemia and should be monitored. MSF recommends alternating an infusion of 5% glucose over 4 hours after the quinine and also in adults giving the quinine in 250 ml of 5% dextrose and 10ml/kg in children.
Special populations:
- Only quinine needs to be dose reduced by one third if there is an acute kidney injury. Those on dialysis do not need a dose adjustment and artemisinin derivatives do not require any dose adjustments.
- Pregnant women have a higher mortality and should receive the parenteral treatment available. Ideally artesunate, then artemether and then quinine. Hypoglycaemia is also more common.
- HIV patient drug interactions: https://www.hiv-druginteractions.org.
Switching to oral:
- If the duration of parenteral treatment was less than 7 days: treat 3 days with an ACT or seven days with quinine.
- If quinine IV has been used and AS-MQ is to be used, an interval of 12 hours should elapse between the last dose of quinine and the administration of MQ.
- If the patient initially presented with impaired consciousness then mefloquine should ideally be avoided due to the increased neuropsychiatric complications.
- When ACT is not available consider artesunate+clindamycin, artesunate + doxycycline, quinine + clindamycin or quinine + doxycycline. Clindamycin is preferred in children (<8 years) and pregnant women.
- Check haemoglobin 14 days after the start of IV treatment to check the extent of potential haemolysis both due to the destruction of infected RBCs and also secondary to the treatment given.
Other Treatments:
- Antipyretics: Paracetamol is still recommend by WHO.
- Blood: transfuse as required:
- Hb <7g/dL in areas of low transmission.
- Hb <4g/dL in high transmission areas unless there are signs of decompensation prior to this.
- If <36/40 weeks pregnant transfuse Hb <7 g/dL.
- If >36/40 weeks pregnant transfuse Hb <8 g/dL.
- If the patient is bleeding then use fresh whole blood or cryoprecipitate, fresh frozen plasma and platelets. Vitamin K can also be administered.
- Glucose: these patients require regular monitoring.
- Anticonvulsants: if two or more seizures occur within 24 hours. This is because seizures from P. falciparum are more common in children but there is also an overlap with febrile convulsions. Therefore 2 seizures within 24 hours constitutes severe malaria and should be treated as such. There is no specific recommendation for one anticonvulsant other another except to say when using anything that can cause respiratory depression, respiratory support/equipment needs to be available as one study showed increased mortality when using phenobarbitone for this very reason.
- Renal failure: may require renal supportive care (haemodialysis, haemofiltration or peritoneal dialysis).
- Broad spectrum antibiotics: by definition of severe malaria it will be impossible to differentiate severe malaria +/- meningitis or septicaemia especially in children. Therefore it is recommended to cover for severe infection and where possible perform a lumbar puncture.
- Fluids: there is no particular recommendations for fluid management in malaria and do whatever is your current practise, as no doubt as long as there are doctors there will be a fluid argument. Importantly if a child has concurrent malnutrition resulting in oedema (kwashiorkor) then there is a very high mortality and risk that fluids will cause heart failure.
Q10. I’m going to a malaria endemic country, how do I protect myself?
Answer and interpretation
Awareness: – see NaTHNac for up to date information specific country malarial risk and resistance if taking prophylaxis. For an in-depth walk through for travellers download the UK guidelines.
Bites by mosquitoes:
• Knock down insecticides (probably not something you will personally have).
• Pyrethroid impregnated nets.
Permithrin impregnated clothing.
• Air conditioning (the life cycle is slower in colder conditions and mosquitoes will avoid these environments).
• Pyrethroid vaporisers.
• Repellents (DEET 20-50% lasts up to 12 hours. Citronella effective for 30mins, Lemon Eucalyptus oil 10-30% – mosiguard natural – is the only proven “natural” repellent lasts 2-5 hours.
Click for Chemoprophylaxis
Diagnose malaria promptly:
Some travellers are doing self rapid tests and only screening once they get a fever and taking treatment for malaria at this point. Highest sensitivity for P. falciparum. (P. vivax 66-88%, P. ovale 5.5-87%, P. malariae 21-45%).
Presumptive anti-relapse therapy (PART):
• 14 days course of primaquine to eliminate potential hypnozoites after return.
• Only appropriate after very high risk exposure in vivax/ovale area.
• Should overlap with final week of standard prophylactic drug.
• See expert opinion/help if doing this.
Summary
- 5 types of malaria infect humans. P. falciparium causes the most morbidity and mortality and is associated with cerebral malaria. Hence one not to miss.
- P. vivax and P. ovale can be dormant in the liver (hypnozoites) and hence months later cause another bout of malaria. You need treat with primaquine to eradicate the hypnozoites.
- Fever in the returned traveller, think malaria and order a thick and thin and rapid diagnostic test (RDT), if negative you are done, think of other causes (unless highly suspicious – repeat).
- Malaria is hard to diagnose in pregnancy as the parasite likes the placenta and reduces test sensitivity. Repeat thick and thin in this scenario and speak to an expert.
- To treat malaria, pretty much everyone (except 1st trimester pregnancy – guidelines likely to change shortly) can be on a 3 day course of ACT – artesunate combination therapy.
- If severe treat everyone with IV artesunate.
- Protect yourself by taking chemoprophylaxis, sleeping under a impregnated bed net (the mosquito bites at dusk and night time), use air conditioning where possible, use mosquito repellant.
The following are useful websites for the traveler and treatment in adults and paediatrics:
- National Travel Health Network and Centre – NaTHNaC.
- Taylor TE. Caring for children with cerebral malaria: insights gleaned from 20 years on a research ward in Malawi.
- WHO – Guidelines for the treatment of malaria.
- WHO – Management of Severe Malaria.
References
- Beeching N and Gill G. Lecture Notes – Tropical Medicine. 7e Wiley Blackwell 2014.
- CDC malaria
- Dondorp A, et al; South East Asian Quinine Artesunate Malaria Trial (SEAQUAMAT) group. Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet. 2005 Aug 27-Sep 2;366(9487):717-25. PubMed PMID: 16125588.
- Dondorp AM, et al; AQUAMAT group. Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): an open-label, randomised trial. Lancet. 2010 Nov 13;376(9753):1647-57. doi: 10.1016/S0140-6736(10)61924-1. Epub 2010 Nov 7. Erratum in: Lancet. 2011 Jan 8;377(9760):126. PubMed PMID: 21062666; PubMed Central PMCID: PMC3033534.
- Eddleston, Davidson, Brent, Wilkinson. Oxford Handbook of Tropical Medicine. Oxford Medical Handbooks. 4e 2014
- Gething et al. A new world malaria map: Plasmodium falciparum endemicity 2010. Malaria Journal 2011
- Guidelines for malaria prevention in travellers from the UK: 2017
- Lancet – Malaria in pregnancy 3
- Lalloo D. UK Malaria treatment guidelines 2016
- Malaria in Pregnancy consortium – MIP
- Matthews PC. Tropical Medicine Notebook. Oxford University Press, 2017
- MSF – Malaria Clinical Guidelines
- National Travel Health Network and Centre – NaTHNaC
- Rothe C. Clinical Cases in Tropical Medicine. Elsevier 2015. ISBN 9780702058240
- Taylor TE. Caring for children with cerebral malaria: insights gleaned from 20 years on a research ward in Malawi.
- WHO – Guidelines for the treatment of malaria
- WHO – Management of Severe Malaria
CLINICAL CASES
Tropical Travel Trouble
Dr Neil Long BMBS FACEM FRCEM FRCPC. Emergency Physician at Kelowna hospital, British Columbia. Loves the misery of alpine climbing and working in austere environments (namely tertiary trauma centres). Supporter of FOAMed, lifelong education and trying to find that elusive peak performance.