Goodbye “Massive” and “Submassive”
The 2026 Acute Pulmonary Embolism (PE) Guidelines Have Arrived
The first-ever AHA/ACC clinical practice guideline on acute pulmonary embolism drops a new A-to-E severity classification, promotes PERT teams to Class 1, and finally says it: DOACs over warfarin, LMWH over UFH. Here’s what emergency physicians need to know…
Creager MA et al. 2026 AHA/ACC/ACCP/ACEP/CHEST/SCAI/SHM/SIR/SVM/SVN Guideline for the Evaluation and Management of Acute Pulmonary Embolism in Adults: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2026 Mar 24;153(12):e977-e1051.
Wait…hasn’t there been an AHA/ACC PE guideline before?
No. Not a formal one.
The 2011 AHA scientific statement on massive and submassive PE was widely used but it was not a clinical practice guideline. The 2019 ESC guidelines from Europe became the de facto standard for many clinicians. This 2026 document is the first dedicated AHA/ACC clinical practice guideline for acute PE in adults. It is a big deal.
What’s the biggest single change?
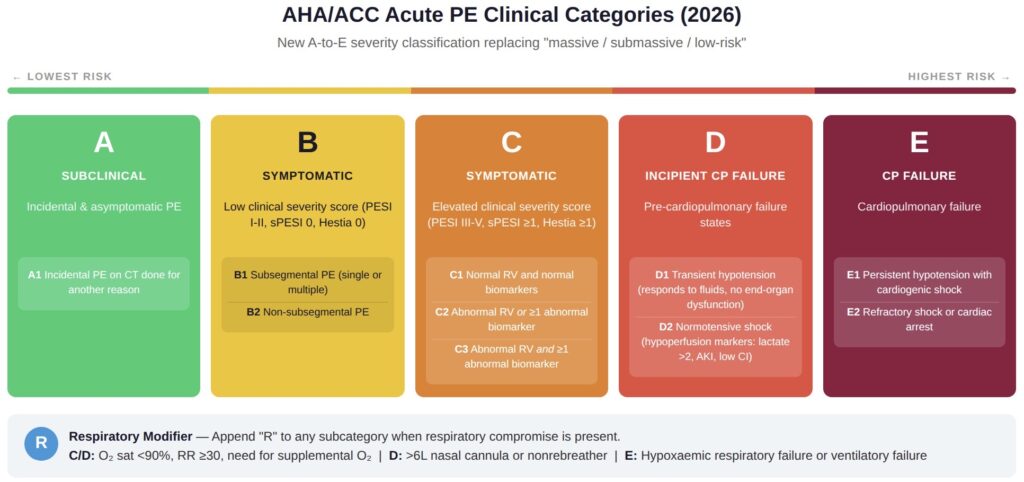
A completely new severity classification. The old language of massive, submassive, and low-risk…is gone. In its place: AHA/ACC Acute PE Clinical Categories A through E, with subcategories.
Think of it as a grading scale:
- Category A: Subclinical. Incidental PE found on CT done for another reason, no symptoms. These patients can go home from the ED.
- Category B: Symptomatic but low clinical severity score (PESI I-II, sPESI 0, Hestia 0). Early discharge is generally recommended.
- Category C: Symptomatic with elevated severity score (PESI III-V, sPESI ≥1, Hestia ≥1). Subcategories C1-C3 further separate patients by RV dysfunction and biomarker elevation.
- Category D: Incipient cardiopulmonary failure. Normotensive shock or pre-failure states. The patient who looks “fine” on the blood pressure cuff but has rising lactate and creatinine.
- Category E: Cardiopulmonary failure. E1 is persistent hypotension with cardiogenic shock. E2 is refractory shock or cardiac arrest.
There is also a respiratory modifier (R) that can be appended to any subcategory when hypoxia, tachypnea, or escalating oxygen requirements are present.
Why did they replace “massive” and “submassive”?
Because the old terms were too blunt.
“Submassive” covered a huge range of patients from the one who is hypoxic and deteriorating, to the one who has a mildly elevated troponin and feels fine. The new system captures that difference. Category C1 (elevated severity score but normal RV and normal biomarkers) is very different from Category D2 (normotensive shock with end-organ dysfunction), and each one now has its own name and its own management path.
What does this mean for my ED workflow?
Quite a lot. Here is the short version:
Category A or B → Think about sending them home. The guideline says outpatient treatment is reasonable for Category A and B patients using a decision tool (Hestia, PESI, or sPESI). This is a Class 2a recommendation backed by randomised trial data. The HOME-PE trial showed both Hestia and sPESI performed equally well for selecting safe-to-discharge patients.
Category C and above → Hospitalise, measure biomarkers, image the RV. At least one cardiac biomarker (troponin or BNP) should be measured. Lactate (venous or arterial) is now a Class 1 recommendation for Categories C-E. And RV imaging — preferably echocardiography over CT — is recommended for risk stratification.
Categories C-E → Activate PERT.
The recommendation for PE response teams is now Class 1, Level B-NR. This is a notable upgrade.

DOACs finally get the crown?
Yes. The guideline states clearly:
In patients with acute PE who are eligible for oral anticoagulation, DOACs are recommended over vitamin K antagonists, unless contraindicated. (Class 1, Level B-R)
And for parenteral anticoagulation:
LMWH is recommended over UFH. (Class 1, Level B-R)
The reasoning: LMWH reduces recurrent VTE more effectively than UFH without increasing major bleeding. It has predictable dosing, does not require routine monitoring, and carries lower risk of heparin-induced thrombocytopenia. DOACs are preferred over warfarin for lower bleeding risk (especially intracranial haemorrhage), simpler dosing, and fewer drug-food interactions.
Any surprises in anticoagulation for special populations?
Several.
Obesity (BMI >30): DOACs are reasonable over warfarin — even in severe obesity. Meta-analyses show apixaban and rivaroxaban are at least as safe and effective as warfarin in this group.
Antiphospholipid syndrome: Warfarin wins here. DOACs are associated with higher arterial thrombotic events in thrombotic APS. The exception: single-antibody, low-risk APS without history of arterial thrombosis — a DOAC may be considered.
Pregnancy: LMWH or UFH only. DOACs and warfarin are classified as potentially harmful (Class 3: Harm).
Kidney disease (stage 2-3): DOAC over warfarin (Class 1, Level A). For stage 4-5 or dialysis, apixaban may be considered but the data are uncertain.
Liver disease: DOACs are reasonable for Child-Pugh A and B. For Child-Pugh C: avoid DOACs (Class 3: Harm).
What about thrombolysis and advanced therapies?
The guideline introduces a structured approach based on the new categories. Here is the practical summary:
- Categories A-C2: Systemic thrombolysis is harmful (Class 3: Harm). CDL and MT are not recommended.
- Category C3: The role of systemic thrombolysis and catheter-based therapies is “unclear.” The PEITHO trial showed thrombolysis prevented cardiovascular collapse but at the cost of more bleeding, including intracranial haemorrhage.
- Categories D1-D2: Advanced therapies “may be considered” — systemic thrombolysis, CDL, or MT.
- Category E1: Advanced therapies are “reasonable” — systemic thrombolysis, CDL, MT, or surgical embolectomy.
- Category E2: Systemic thrombolysis is reasonable. Surgical embolectomy is not recommended over other options like VA-ECMO.
One important nuance: the PEERLESS trial (550 patients randomised to CDL vs MT) showed no significant difference in 30-day mortality or major bleeding between the two approaches. So if a catheter-based approach is selected, the choice between CDL and MT remains operator- and situation-dependent.
What about sedation. Is that really in the PE guideline?
Yes, and for good reason.
The guideline includes a Class 3: Harm recommendation against deep sedation and mechanical ventilation unless clinically necessary in Categories C-E. Case series document cardiac arrest rates of 19-28% after anaesthesia induction in patients with PE and RV dysfunction, even in those who were haemodynamically stable beforehand. Sedation blunts the compensatory sympathetic response that is keeping these patients alive. If intubation is required, vasopressors, inotropes, and VA-ECMO should be ready at the bedside.
What about IVC filters?
The guideline is clear: no routine IVC filter placement in anticoagulated patients (Class 3: Harm, Level A). Filters are reserved for patients who genuinely cannot receive anticoagulation. Retrievable filters are preferred over permanent ones, and the FDA recommends retrieval within 29-54 days. A structured follow-up programme is recommended to improve retrieval rates.
What should I do differently in my next shift?
- Start thinking in A-E categories rather than massive/submassive/low-risk.
- Measure lactate in all patients with Category C-E PE. It is now a Class 1 recommendation.
- Use a decision tool (Hestia, PESI, sPESI) to identify which patients can safely go home.
- Order echocardiography (preferred over CT) for RV assessment in Category C-D patients.
- Start LMWH rather than UFH drip if parenteral anticoagulation is needed.
- Activate PERT early for Categories C-E. It is now formally recommended as Class 1.
- Be cautious with sedation in any patient with suspected RV dysfunction.
What about follow-up?
The guideline recommends clinical follow-up within the first week of discharge, a dedicated visit at or before 3 months, and screening for PE-related symptoms and functional limitations at every visit for at least one year. Follow up is aimed specifically to catch chronic thromboembolic pulmonary disease (CTEPD), which complicates roughly 3% of acute PEs.
For patients on extended anticoagulation (beyond 3-6 months): half-dose apixaban (2.5 mg twice daily) or rivaroxaban (10 mg daily) is now recommended over full-dose to reduce bleeding while maintaining protection against recurrence. This is supported by the recent RENOVE and API-CAT trials.
Iterative learning tool
We have designed a tool as a formative feedback loop to help readers identify knowledge gaps and asses information retrieval. Still in early stages, but would be great if you could check it out. Feedback appreciated:
Test Yourself: USMLE-Style Questions
Ready to check if these new guidelines have stuck?
Try these six questions complete with answers and explanations.
References
- Creager MA et al. 2026 AHA/ACC/ACCP/ACEP/CHEST/SCAI/SHM/SIR/SVM/SVN Guideline for the Evaluation and Management of Acute Pulmonary Embolism in Adults: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2026 Mar 24;153(12):e977-e1051.
- Long B. 2026 Guideline: Evaluation and Management of Acute PE. emDocs
Internist at the Medical University of Vienna and founder of Medmastery. Master’s degree in public health at Johns Hopkins University as a Fulbright student. Passionate about teaching. | Medmastery | LinkedIn | Twitter |
BA MA (Oxon) MBChB (Edin) FACEM FFSEM. Emergency physician, Sir Charles Gairdner Hospital. Passion for rugby; medical history; medical education; and asynchronous learning #FOAMed evangelist. Co-founder and CTO of Life in the Fast lane | On Call: Principles and Protocol 4e| Eponyms | Books |

