History of fluorescein
Fluorescein is a fluorescent dye used to make otherwise subtle ocular pathology visible. On the ocular surface it highlights epithelial barrier disruption and tear-film patterns; elsewhere it underpins tests for aqueous leakage (Seidel), lacrimal drainage (Jones), and retinal vascular imaging (fluorescein angiography).
Fluorescein basics
Fluorescein is a synthetic xanthene dye (first synthesised by Adolf von Baeyer in 1871) most commonly used clinically as sodium fluorescein (C₂₀H₁₀Na₂O₅) on sterile strips or as solution. When excited by blue light, it emits bright green/yellow-green fluorescence (blue excitation ~494 nm; green emission ~521 nm), which is why cobalt-blue illumination is so useful at the slit lamp.
How it works on the ocular surface
Fluorescein highlights epithelial compromise because a damaged surface cannot exclude water-soluble dye as effectively as intact epithelium. Clinically, this makes abrasions, ulcers, dendrites and contact lens–related staining patterns easier to detect and document.
Modern interpretation of “staining”
Clinical staining is best understood as a test of epithelial barrier function, not simply a marker of “missing epithelium”. Water-soluble dyes are normally excluded by tight junctions and the apical glycocalyx, while shed or degenerating cells can take up dye. Sparse punctate fluorescein uptake can occur even on some normal corneas and may reflect graded loss of glycocalyx barrier permitting dye entry into “pre-shed” cells. The traditional explanation of dye “micropooling” in tiny epithelial defects is likely over-simplified; the term “punctate epithelial erosions” may therefore be a misnomer.
Why cobalt blue matters
Fluorescein can be seen in white light, but fluorescence makes interpretation dramatically easier. Cobalt-blue excitation boosts signal and suppresses background glare, turning subtle staining into a readable pattern (especially in contact lens fitting and micro-abrasions).
Common clinical uses
Common ocular surface diagnoses where fluorescein is useful include superficial punctate keratitis/dry eye, corneal abrasion or erosion, foreign body with linear tracking patterns, herpetic keratitis, corneal ulceration, epithelial basement membrane dystrophy, and contact lens–related epithelial disruption.
Fluorescein is used for more than “abrasions and ulcers”, including:
- Corneal damage and dry eye (punctate staining patterns, dendrites, abrasions, ulcers)
- Aqueous leak / open globe assessment (Seidel test “waterfall/streaming” washout sign)
- Lacrimal drainage testing (Jones I / nasolacrimal duct obstruction workup)
- Retinal vascular imaging (fundus fluorescein angiography)
Vital stains at a glance
Differences between fluorescein, lissamine green and rose bengal reflect molecular size/charge and visibility under different illumination, with fluorescein tending to spread more rapidly from punctate sites.
| Dye | Best for | What it highlights | Practical notes |
|---|---|---|---|
| Sodium fluorescein | Cornea / tear film | Epithelial barrier compromise; tear film patterns | View with blue excitation (cobalt blue). Tear film fluorescence can obscure staining if assessed too early. |
| Rose bengal | Conjunctiva / ocular surface disease | Devitalised/compromised surface and mucus (classically KCS patterns) | Historically important; often uncomfortable |
| Lissamine green | Conjunctiva / ocular surface disease | Degenerate cells, dead cells and mucus; similar pattern to rose bengal | Norn: suitable for cornea and conjunctiva; “no irritation” and no side-effects detected in his series. |
History
1871 – Adolf von Baeyer synthesises fluorescein (“resorcinphthalein”), creating the dye scaffold that later becomes central to ophthalmic diagnostics.
1882 – Paul Ehrlich introduced fluorescein as an experimental diagnostic tracer in ophthalmology (“Fluorescein methode”) and described provoked ocular fluorescence in rabbits after fluorescein administration. He described a distinct corneal fluorescent “line” which was visible even when the cornea appears otherwise colourless and demonstrated fluorescein could reveal fluid emerging from the posterior chamber after corneal puncture.
1882 – Ernst Pflüger uses fluorescein as a physiological tracer in rabbits in Zur Ernährung der Cornea. After instilling a succinyl fluorescein solution into the conjunctival sac, he notes that within minutes the corneal surface fluoresces green (faintly but clearly). By creating fine epithelial defects he observed characteristic advancing fluorescent patterns from the limbus toward the centre. He interpreted these as evidence of a centripetal stromal fluid stream and proposed that corneal nutrition depends primarily on surrounding vascular tissues rather than chiefly on aqueous humour.
Träufelt man einem Kaninchen einige Tropfeneiner 1/4%igen Lösung dieses Succinylfluoresceïns, in den Conjunctivalsack, so sieht man nach einigen Minuten die Oberfläche der Cornea wenig intensiv aber deutlich erkennbar in grüner Farbefluoresciren – Pflüger, 1882
If you drip a rabbit’s conjunctival sac with a few drops of a 0.25% solution of this succinyl fluorescein, then after a few minutes you see the surface of the cornea fluoresce in green—not very intensely, but clearly recognisable – Pflüger, 1882
1888 – Manuel Straub demonstrates that fluorescein rapidly distinguishes intact cornea (minimal dye uptake) from epithelial loss (immediate intense green staining). He notes that a drop of concentrated fluorescein in 1% soda solution stains the floor of a corneal ulcer or epithelial defect bright green, then fades within about an hour.
In a clinical case after scalding spirit vapour, both corneas appeared clear on inspection, but fluorescein instantly revealed diffuse green staining of the injured cornea, confirming epithelial loss when other methods could not. Using serial staining to track epithelial regeneration day by day, Straub postulates that the dye does not penetrate living cells; rather it passes through the intercellular “cement substance”, explaining why uptake is so rapid only when the epithelial barrier is disrupted.
Ein Tropfen der Fluorescinlösung brachte sofort Klarheit. Während an dem rechten Auge der Farbstof nirgendwo eindrang, zeigte die ganze Hornhaut des linken Auges gleich nach der Einträufelung eine stark grüne Verfärbung, so dass die Diagnose Epithelverlust damit begründet erschien. Straub 1888
A single drop of fluorescein solution brought immediate clarity: the right eye took up no dye, while the entire cornea of the left eye turned intensely green straight after instillation—supporting the diagnosis of epithelial loss. Straub 1888
1891 – Fromm & Groenouw refine fluorescein staining into a reproducible clinical method. They proposed the use of 2% potassium fluorescein solution with eyelids closed for up to 30 seconds if needed and recommended to avoid high concentrations that could artefactually stain intact epithelium. They observed that fluorescein appears green on cornea but yellow on conjunctiva because the cornea is seen against a darker background (reflected light), whereas conjunctiva is viewed with transmitted light reflected off white sclera.
1899 – W. Bihler extends fluorescein from epithelial defects to a proposed method for detecting endothelial Descemet’s changes in early sympathetic ophthalmia and inflammatory states. He popularising an explicit “recipe”: cocainise the eye, instil 5% potassium fluorescein (with 1–2% soda), and rinse after 30 seconds; he notes intact cornea may take ~15 minutes for flecks, whereas endothelial changes show earlier green staining.
1902 – Arthur H. Benson (Dublin) publishes A note on the value of the fluorescein test, summarising fluorescein salt forms in use (sodic/“uranin” vs potassium) and their intense yellow-green fluorescence. He argued against simplistic textbook rules that “only ulcers stain” and proposed that staining may extend beyond visible ulcer margins; epithelium damaged by heat or chemicals can stain without abrasion; and that heavy cocaine use can produce mottled staining by reducing epithelial vitality.
1920 – Erich Seidel shows that topical 2% fluorescein can reveal an otherwise invisible aqueous leak after Elliot trephination. A weak green-yellow tint over the trephination site rapidly becomes an almost white track that runs vertically inferiorly within seconds, as clear aqueous dilutes/clears the dye, creating the recognisable “streaming/washout” sign (illustrated in Abb. 1–4). He notes that without fluorescein the outflow could not be detected even with slit-lamp magnification, and describes the phenomenon as a continuous aqueous stream that becomes visible only because it alters the fluorescein colour/intensity along its course.
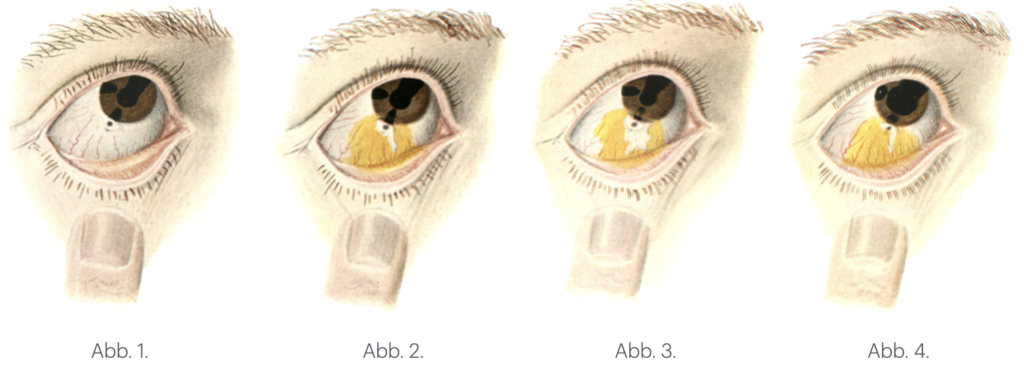
Abb. 1: “Before fluorescein (no visible leak)”
Abb. 2: “Fluorescein applied at trephination site”
Abb. 3: “Positive Seidel: aqueous ‘washout’ begins”
Abb. 4: “Streaming track to inferior fornix (classic sign)”
1921 – Seidel formalises his findings into a practical test of filtration (“Fistelprobe”) for scleral scars after successful Elliot trephination to demonstrate a functioning communication between the anterior chamber and subconjunctival space. The same optical phenomenon was later generalised beyond glaucoma surgery to the modern Seidel test and is used for corneal perforation, post-operative wound leaks, and suspected open globe injury.
1933 – Henrik Sjögren introduces rose bengal for ocular surface staining (particularly conjunctiva/tear film disease), shifting conjunctival assessment away from fluorescein-alone.
1938 – The turning point for the use of fluorescein in ophthalmology. Theodore Ernst Obrig noted that judging limbal clearance when fitting a contact lens was “practically impossible” under the intense white beam of slit illumination due to surface reflections from the lens, sclera and iris. Obrig placed a cobalt-blue filter between the light source and the eye, and added a buffer solution containing one drop of fluorescein to a contact lens before insertion. He noted that the fluorescein glowed brilliantly in blue light with background glare suppressed. noted that cobalt-blue light was originally intended for examining scleral and conjunctival vessels, but is ideal for contact lens fitting and highlighting minute corneal abrasions.
1938 – A turning point in practical fluorescence. Theodore Ernst Obrig showed that judging limbal and per-limbal clearance during contact lens fitting was “practically impossible” under the intense white beam of slit illumination because of surface reflections from the lens, sclera and iris. He placed a cobalt-blue filter between the light source and the eye and added a buffer solution containing one drop of fluorescein to the lens before insertion. Under cobalt-blue illumination he observed the fluorescein “glow brilliantly” while background glare was suppressed with yellow-green indicating non-contact, and dark areas indicating contact. Obrig also found that the cobalt-blue spotlight, originally intended for examining scleral and conjunctival vessels, proved ideal for contact lens fitting and for detecting minute corneal abrasions.

1961 – Harold R. Novotny and David L. Alvis published a practical method for studying retinal blood flow in humans using intravenous fluorescein and serial fundus photography. In their Circulation report they describe a Zeiss fundus camera with electronic flash and define the optical requirements for fluorescein imaging (blue excitation around 490 nm and green emission around 520 nm). In a darkened room they rapidly injected 5 mL of 5% fluorescein in sodium bicarbonate (Fluorescite) into an antecubital vein, and photograph the retinal circulation at 12-second intervals for ~3½ minutes. This simple method captured distinct arteriolar and venous filling phases and revealed vascular detail not readily seen with routine ophthalmoscopy or standard fundus photographs.
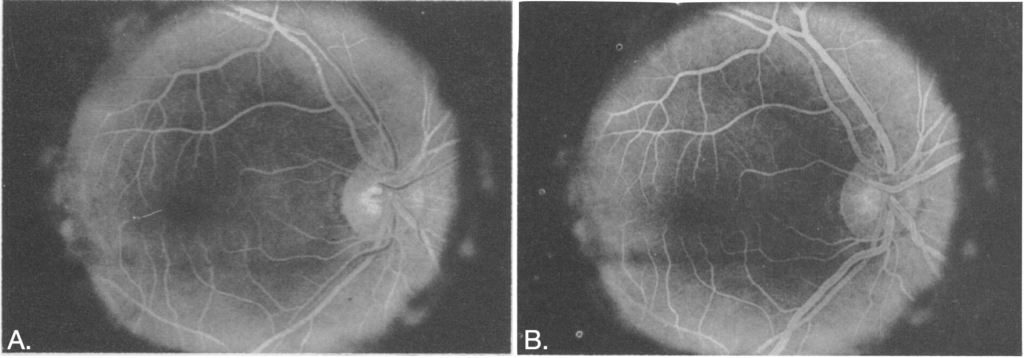
Novotny Alvis Photographing Fluorescence in Circulating Blood in the Human Retina 1961
Later accounts note the technique was first developed in 1959 while they were medical students, trialled initially in rabbits, and that the first successful human angiogram was performed on Alvis after a coin toss.
1972 – Lester Tallmon Jones et al quantify lacrimal drainage using a primary dye (fluorescein) conduction test“ in healthy volunteers. With either the superior or inferior canaliculus occluded, they instil one drop of 2% fluorescein and begin sampling the inferior nasal meatus 30 seconds later, reinserting the cotton applicator at 30-second intervals to record time-to-first appearance and dye quantity. Their results show broadly similar performance of the two canaliculi supporting the practical inference that loss of either canaliculus alone usually won’t materially impair tear drainage.
1973 – Mogens Stig Norn introduces lissamine green as a practical ocular-surface vital dye in Acta Ophthalmologica. Studying 171 eyes by slit lamp and microscopy of stained specimens, he shows lissamine green stains degenerate cells, dead cells and mucus, with vital-staining properties “almost identical” to rose bengal, and notes that instillation causes no irritation with no side-effects detected.
He also demonstrates that combining lissamine green with rose bengal produces a stable blue vital stain, and warns that adding fluorescein can suppress the green component—useful practical guidance when clinicians mix dyes.
1977 – Lester T. Jones describes a painless, gentle lacrimal fluorescein test for routine diagnosis of lacrimal obstruction. He instructs clinicians to instil a small drop of fluorescein in each eye and not wipe the tears, then anaesthetise the anterior end of the inferior turbinate with 2% cocaine, and gently pass a small cotton-tipped applicator into the anterior inferior nasal meatus; fluorescein staining of the applicator can be detected within five minutes. He notes that the abnormality causing epiphora often involves the anterior end of the inferior turbinate, and emphasises atraumatic technique.
Associated Persons
- Johann Friedrich Wilhelm Adolf von Baeyer (1835–1917) – Germany; chemist. Synthesised fluorescein (1871), establishing the parent xanthene dye later adopted in ophthalmic diagnostics.
- Paul Ehrlich (1854–1915) – Germany; physician-scientist. Introduced fluorescein as an ophthalmic tracer in rabbit experiments (1882), proposing human applicability.
- Ernst Pflüger (1846–1903) – Switzerland; ophthalmologist. Used fluorescein as a physiological tracer in rabbits (1882), demonstrating green corneal fluorescence and centripetal “flow” patterns after epithelial disruption.
- Manuel Straub (1858–1916) – Netherlands; ophthalmologist (Amsterdam). Demonstrated clinical fluorescein for corneal epithelial loss (1888) and used it to follow epithelial regeneration.
- Erich Seidel (1882–1946) – Germany; ophthalmologist (Heidelberg). Described fluorescein “washout/streaming” after trephination (1920) and formalised the filtration “Fistelprobe” (1921).
- Henrik Sjögren (1899–1986) – Sweden; ophthalmologist. Introduced rose bengal for keratoconjunctivitis sicca assessment (1933).
- Theodore Ernst Obrig (1894-1967) – USA; contact lens manufacturer/optician. Formalised cobalt-blue excitation of fluorescein for contact lens fitting (1938).
- Mogens Stig Norn (1925–2023) – Denmark; ophthalmologist (Copenhagen). Introduced lissamine green for vital staining of cornea/conjunctiva (1973)
- Lester Tallmon Jones (1894-1983) – USA; ophthalmologist (Portland). Quantified fluorescein dye transit to the inferior meatus (1972) and described a gentle office-based lacrimal fluorescein test for obstruction (1977)
References
Historical references
- Kekuié, August. Untersuchungen über aromatische Verbindungen Ueber die Constitution der aromatischen Verbindungen. I. Ueber die Constitution der aromatischen Verbindungen. 1866; 137(2): 129–196
- Baeyer A. Ueber eine neue Klasse von Farbstoffen. Berichte der deutschen chemischen Gesellschaft, 1871; 4(2): 555–558
- Ehrlich P. Ueber provocirte Fluorescenzerscheinungen am Auge. Deutsche Medizinische Wochenschrift. 1882; 8: 54–55
- Pflüger E. Zur Ernährung Der Cornea. Klinische Monatsblätter für Augenheilkunde, 1882; 20: 69–82
- Straub M. Fluoreszeinlösung als ein diagnostisches Hilfsmittel für Hornhauterkrankungen. Centralblatt für Augenheilkunde. 1888; 12: 75–77
- Fromm, Groenouw. Ueber die diagnostische Verwendbarkeit der Fluoresceinfärbung bei Augenerkrankungen. Archiv für Augenheilkunde. 1891; 22: 247–257.
- Bihler W. Zur Diagnose von Endothelerkrankungen der Hornhaut mittelst Fluorescein, insbesondere bei beginnender sympathischer Ophthalmie. Münchener Medizinische Wochenschrift. 1899; 46: 1045–1048
- Benson AH. A note on the value of the fluorescein test. Ophthalmic Review. 1902;21:121–130
- Seidel E. Weitere experimentelle Untersuchungen über die Quelle und den Verlauf der intraokularen Saftströmung. III. Mitteilung. Über den Vorgang der physiologischen Kammerwasserabsonderung und seine pharmakologische Beeinflussung. Albrecht von Graefes Archiv für Ophthalmologie 1920; 102: 366–382
- Seidel E. Weitere experimentelle Untersuchungen über die Quelle und den Verlauf der intraokularen Saftströmung. VI. Mitteilung. Die Filtrationsfähigkeit, eine wesentliche Eigenschaft der Scleralnarben nach erfolgreicher Elliotscher Trepanation. Albrecht von Graefes Archiv für Ophthalmologie 1921; 104: 158–161
- Sjögren H. Zur Kenntnis der keratoconjunctivitis sicca. Keratitis filiformis bei Hypofunktion der Tränendrüsen. Acta Ophthalmol Supplementum 1933; 2: 1–151. Doctoral thesis, Karolinska Institutet, Stockholm
- Obrig TE. A Cobalt Blue Filter for Observation of the Fit of Contact Lenses. Arch. Ophth. 1938; 20: 657
- Obrig TE. Contact lenses. 1942
- Novotny HR, Alvis DL. A method of photographing fluorescence in circulating blood in the human retina. Circulation. 1961 Jul;24:82-6
- Jones LT, Marquis MM, Vincent NJ. Lacrimal function. Am J Ophthalmol. 1972 May;73(5):658-9.
- Norn MS. Lissamine green. Vital staining of cornea and conjunctiva. Acta Ophthalmol (Copenh). 1973;51(4):483-91.
- Jones LT. Lacrimal fluorescein test. Am J Ophthalmol. 1977 May;83(5):762.
- Alvis DL, Julian KG: The story surrounding fluorescein angiography. Journal of Ophthalmic Photography 1982; 5(1): 6-8
Eponymous term review
- Riggs JL, Seiwald RJ, Burckhalter JH, Downs CM, Metcalf TG. Isothiocyanate compounds as fluorescent labeling agents for immune serum. Am J Pathol. 1958 Nov-Dec;34(6):1081-97.
- Norn MS. Vital staining of cornea and conjunctiva. Acta Ophthalmol (Copenh). 1962;40:389-401
- Cain W Jr, Sinskey RM. Detection of anterior chamber leakage with Seidel’s test. Arch Ophthalmol. 1981 Nov;99(11): 2013.
- Saine PJ. Landmarks in the historical development of fluorescein angiography. J Ophthalmic Photogr. 1993 Apr;15(1):17-23.
- Carter Ramsey A. Vital Stains: What You Really Need to Know. Review of cornea and contact lenses. April 18, 2011
- Bron AJ, Argüeso P, Irkec M, Bright FV. Clinical staining of the ocular surface: Mechanisms and interpretations. Prog Retin 2015;44:36-61
- Cooksey CJ. Quirks of dye nomenclature. 9. Fluorescein. Biotech Histochem. 2017;92(7):506-512.
- Sauer M. Fluorescein. Moran Core. 2020
- Naik V. A brief history of fluorescein. Hektoen International 2025
- Martin A. Corneal staining with fluorescein. J Med Insight. 2024;2024(395)
eponymictionary
the names behind the name
BVisSci, MD, University of Notre Dame, Fremantle. Doctor, baker, avid traveller, aspiring ophthalmologist
BA MA (Oxon) MBChB (Edin) FACEM FFSEM. Emergency physician, Sir Charles Gairdner Hospital. Passion for rugby; medical history; medical education; and asynchronous learning #FOAMed evangelist. Co-founder and CTO of Life in the Fast lane | On Call: Principles and Protocol 4e| Eponyms | Books |

