Paracetamol or Acetaminophen?
Paracetamol and acetaminophen are not two different drugs but two later names for an old compound whose history runs in three stages: it was first made in the laboratory, then eclipsed by acetanilide and phenacetin, and only later rediscovered as the key metabolite linking those earlier drugs to modern practice.
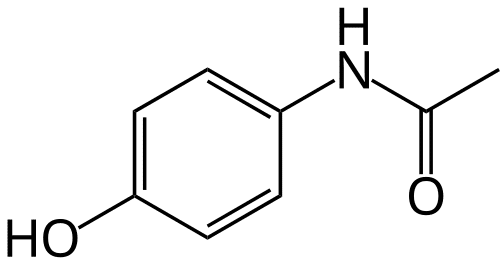
Although perhaps we should all be calling it N-acetyl-p-aminophenol, for the purposes of this article, we will refer to this medication as paracetamol.
Original Synthesis
The history of paracetamol begins not in the clinic, but in the chemistry laboratory.
In 1878, the American chemist Harmon Northrop Morse (1848-1920), working at Johns Hopkins University, published Ueber eine neue Darstellungsmethode der Acetylamidophenole (“On a New Method for the Preparation of Acetylamidophenols”). During these experiments, he observed that instead of the expected acetate salt of amidophenol, an acetylamidophenol had formed.
Morse then described the para compound as crystallising in large white prisms, readily soluble in hot water and alcohol, with a melting point of about 179°C. In modern terms, this was paracetamol, chemically characterised long before it was clinically understood. Morse did not investigate any physiological or medicinal effects. At this stage, paracetamol was simply a laboratory compound, made and described, then left without a clinical future for several decades.
The detour through acetanilide and phenacetin
Paracetamol did not move directly from Morse’s bench to the bedside. Instead, the story took a detour through the first generation of synthetic antipyretics.
In 1886, Arnold Cahn and Paul Hepp, working in the Strasbourg clinic of Adolph Kussmaul, published Das Antifebrin, ein neues Fiebermittel (“Antifebrin, a new fever remedy”). Their paper did not describe paracetamol, but acetanilide which they renamed Antifebrin. They reported antipyretic effects in animal experiments and in 24 febrile patients, presenting the drug as a powerful and inexpensive rival to Antipyrin (phenazone), the leading synthetic antipyretic of the day
The famous origin story needs caution. Cahn and Hepp state that “a fortunate accident” placed the preparation in their hands, but the paper itself does not tell the later elaborated tale of intestinal worms, naphthalene, and a pharmacy mix-up. The Strasbourg episode is therefore best understood as an accidental discovery whose details were expanded in later retellings. In retrospect, part of acetanilide’s clinical activity reflected metabolism to paracetamol, but Cahn and Hepp neither discovered paracetamol as a drug nor understood that metabolic relationship.
Acetanilide’s success also exposed the limits of this first generation of synthetic antipyretics. Cahn and Hepp noted cyanosis in some patients, a finding that “at first made them somewhat uneasy,” though they ultimately dismissed it. Later generations would recognise this as an early warning of the haematological toxicity that helped drive the search for supposedly safer related compounds.
That search led to phenacetin (acetophenetidin), developed by Otto Hinsberg with the pharmacologist Alfred Kast as a successor that appeared to preserve the antipyretic and analgesic benefits of acetanilide with fewer obvious side effects. It also fitted the industrial chemistry of the moment. Phenacetin could be made cheaply from para-nitrophenol, a by-product of the German dye industry. Later accounts disagreed on whether its discovery was chiefly a matter of luck or systematic design, but either way phenacetin became a major commercial success. Only in hindsight was it understood that much of its clinical effect led back to paracetamol.
In historical terms, this was the crucial detour. Paracetamol did not move directly from Morse’s bench to the bedside, but re-entered medicine only through the rise and problems of acetanilide and its successors.
Rediscovery of Paracetamol
Paracetamol did not first enter therapeutics in the late 1940s. It had already been clinically examined in the 1890s by Joseph von Mering, who was exploring relationships between chemical structure and antipyretic action among aromatic compounds. In his 1893 review, von Mering traced some of acetanilide’s action to excretion as (acetyl-)paramidophenol, then tested p-amidophenol, acetylamidophenol, and related derivatives directly. He found that acetylamidophenol had prompt antipyretic and antineuralgic effects, but appeared to retain the troublesome adverse effects of the parent amidophenol, including cyanosis and methaemoglobin-related toxicity. By contrast, he described phenacetin as an excellent antipyretic and analgesic with fewer side effects. In practical terms, paracetamol was not embraced as a new wonder drug in 1893 but was examined and passed over.
That early judgement helps explain why phenacetin, rather than paracetamol, dominated the first half of the twentieth century. Later writers have sometimes tried to read more into the 1890s than the evidence allows. Hinsberg’s 1913 retrospective is valuable because it shows that he and later Gustav Treupel were systematically exploring p-aminophenol derivatives, but it does not show that they had already established, in modern metabolic terms, that phenacetin’s effects were mediated mainly through paracetamol. At most, the work of the 1890s suggested a family resemblance among these compounds, not the decisive metabolic explanation that came later.
The real turning point came in the late 1940s. David Lester and Leon A. Greenberg showed that paracetamol occupied a key place in the metabolism of acetanilide and did not reproduce the marked methaemoglobinaemia associated with the older aniline analgesics. Soon afterwards, Brodie and Axelrod demonstrated that a major fraction of phenacetin administered to humans was converted to paracetamol. Paracetamol therefore re-emerged not as a brand-new molecule, but as the previously overlooked compound at the centre of the older drugs’ pharmacology.
That solved the pharmacological story. The naming story was messier.
Why the names split
The two names did not arise because chemists discovered two different drugs, nor because naming authorities deliberately set out to create a transatlantic divide. They arose because the same molecule could be shortened in two different ways from its chemical name, N-acetyl-p-aminophenol and para-acetyl-amino-phenol at a time when nonproprietary drug nomenclature was yet to be formally standardised. The split was therefore commercial first, regulatory second.
1951 – McNeil Laboratories in the United States, began developing the drug with Robert McNeil coining the name acetaminophen from N-acetyl-p-aminophenol. The brand name Tylenol was derived from the same chemical name, and the product was launched in 1955 as Children’s Tylenol Elixir.
1956 – In the United Kingdom, the drug entered clinical use in 1956 through Frederick Stearns & Co., which used the alternative shortening paracetamol from para-acetyl-amino-phenol and marketed it as Panadol.
By the time formal naming systems matured, both names were already established in different markets. Paracetamol became the International Nonproprietary Name (INN) of the World Health Organisation, while acetaminophen remained the United States Adopted Name (USAN). What looks like scientific inconsistency is really the fossil of an earlier era of pharmaceutical marketing and nomenclature.
What should we really call it?
Chemically: N-(4-hydroxyphenyl)acetamide
In US practice: acetaminophen
In international medical writing: paracetamol
For LITFL: paracetamol (acetaminophen) on first mention, then paracetamol
| Term | Meaning / expansion | Status / where used |
|---|---|---|
| Paracetamol | shortened from para-acetyl-amino-phenol | INN International Nonproprietary Name |
| Acetaminophen | shortened from N-acetyl-p-aminophenol | USAN United States Adopted Name |
| APAP | abbreviation of N-acetyl-para-aminophenol | Common abbreviation, especially in US toxicology and labelling |
| N-(4-hydroxyphenyl)acetamide | systematic chemical name | Modern chemistry IUPAC-style naming; also listed by NCI and PubChem |
Associated persons
- Harmon Northrop Morse (1848-1920) – American chemist who first clearly described the synthesis and properties of paracetamol in 1878, long before it became a clinical drug.
- Arnold Cahn (1858–1927) – German internist, surgeon, and paediatrician; with Paul Hepp, identified the antipyretic action of acetanilide (Antifebrin) in Strasbourg in 1886
- Paul Hepp (1851–1912) – German physician and assistant in Strasbourg; co-author with Arnold Cahn of the 1886 paper that introduced acetanilide/Antifebrin as a fever remedy
- Otto Hinsberg (1857–1939) – German chemist who developed phenacetin and later defended the view that its discovery arose from systematic work on aminophenol derivatives rather than mere luck.
- Gustav Treupel (1867–1926) – German internist and university teacher; collaborated in the physiological study of p-aminophenol derivatives and later directed the medical clinic at Frankfurt’s Hospital zum Heiligen Geist
- Joseph von Mering (1849–1908) – German physician and pharmacologist who clinically tested p-amidophenol and acetylamidophenol in the 1890s, helping explain why phenacetin, rather than paracetamol, initially gained favour. He is also remembered for work on diabetes and barbiturates.
- David Lester (1916–1990) – American biochemist; with Leon A. Greenberg, helped establish paracetamol as a key metabolite of acetanilide in the late 1940s.
- Leon A. Greenberg (1907–1985) – American physiologist/pharmacologist at Yale; co-authored the acetanilide metabolism studies with David Lester and is also known for inventing the Alcometer.
- Bernard Beryl Brodie (1907–1989) – British-born American biochemist and pharmacologist; a pioneer of drug metabolism who, with Axelrod and colleagues, showed that much of phenacetin’s action was mediated through paracetamol
- Julius Axelrod (1912–2004) – American biochemist and pharmacologist; co-author of the late-1940s phenacetin metabolism work that helped rehabilitate paracetamol, later awarded the 1970 Nobel Prize for work on neurotransmitters
References
Historical articles
- Gerhardt C. Recherches sur les acides organiques anhydres. Annales de chimie et de physique 1853; 7: 285–342.
- Morse HN. Ueber eine neue Darstellungsmethode der Acetylamidophenole. Berichte der Deutschen Chemischen Gesellschaft 1878; 11(1): 232–233
- Cahn A, Hepp P. Das Antifebrin, ein neues Fiebermittel. Centralblatt für Klinische Medizin, 1886; 7: 561–564.
- von Mering J. Beiträge zur Kenntniss der Antipyretica. Therapeutische Monatshefte, 1893; 7: 577–587.
- Hinsberg O, Treupel G. Ueber die physiologische Wirkung des P-Amidophenols und einiger Derivate desselben. Archiv für Experimentelle Pathologie und Pharmakologie 1894; 33; 216–250.
- Hinsberg, O. Zur Geschichte der Entdeckung der synthetischen Antipyretica. Angewandte Chemie 1913; 26: 148-159
- Gross M. Acetanilid, a critical bibliographic review. New Haven: Hillhouse Press, 1946.
- Lester D, Greenberg LA. The metabolic fate of acetanilid and other aniline derivatives; major metabolites of acetanilid appearing in the blood. J Pharmacol Exp Ther. 1947 May;90(1):68-75.
- Brodie BB, Axelrod J. The estimation of acetanilide and its metabolic products, aniline, N-acetyl p-aminophenol and p-amino-phenol, free and total conjugated, in biological fluids and tissues. J Pharmacol Exp Ther. 1948 Sep;94(1):22-8.
Review articles
- Rainsford KD. Aspirin and Related Drugs. 2004.
- Sneader W. Drugs discovered through serendipity in the laboratory. In: Drug discovery: a history 2005: 438-439
- Nnadi CO, Agbo MO, Uzor PF, Ugwu LO. Development of Differential Spectrophotometric Method for Assay of Paracetamolin Pure and Tablet Dosage Forms. Indian Journal of Pharmacy Research, 2013; 1(1):15-21.
- Brune K, Renner B, Tiegs G. Acetaminophen/paracetamol: A history of errors, failures and false decisions. Eur J Pain. 2015 Aug;19(7):953-65.
- Karet GB. How Do Drugs Get Named? AMA Journal of Ethics. 2019
- Lee WM. Acetaminophen Toxicity: A History of Serendipity and Unintended Consequences. Clin Liver Dis (Hoboken). 2020 Oct 7;16(Suppl 1):34-44. Erratum in: Clin Liver Dis (Hoboken). 2021 Feb 28;17(2):96.
- Rumack BH. Acetylcysteine Treatment of Acetaminophen Overdose: Foundational and Clinical Development. Livers. 2025; 5(2):20.
- National Library of Medicine. “Biographical Overview” (Julius Axelrod profile). Available at: https://profiles.nlm.nih.gov/spotlight/hh/feature/biographical-overview
- The Dispensatory of the United States of America. 1960; 2: . Available at: https://archive.org/details/in.ernet.dli.2015.109419/page/n5/mode/2up?q=1955
- Merriam-Webster. “Paracetamol.” Available at: https://www.merriam-webster.com/dictionary/paracetamol#h1
- Paracetamol. British Pharmaceutical Codex. 1963: 564
- Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (updated 2023) Available at: https://iupac.qmul.ac.uk/BlueBook/PDF/
Hello, my name is…
[etymologically]
MBBS, Curtin University. Junior doctor in the Northern Territory (Royal Darwin Hospital). Interested in physiology, procedures, medical history and fiction. Always careful to check my blind spot when merging into the fast lane.
BA MA (Oxon) MBChB (Edin) FACEM FFSEM. Emergency physician, Sir Charles Gairdner Hospital. Passion for rugby; medical history; medical education; and asynchronous learning #FOAMed evangelist. Co-founder and CTO of Life in the Fast lane | On Call: Principles and Protocol 4e| Eponyms | Books |


Güzel bir yazı olmuş, teşekkürler. Özellikle insightful approach hoşuma gitti.