Cardiovascular Physiology Overview
OVERVIEW
- summary of cardiac muscle, electrical properties, haemodynamics and blood supply
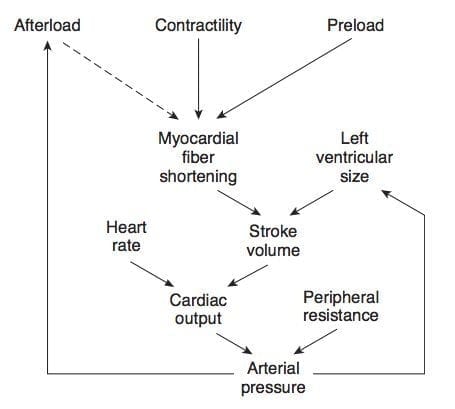
- preload = LV wall stress and end diastole and reflects maximal length of the ventricular sarcomeres when it has filled (just prior to contraction); usually considered the end-diastolic pressure in clinical practice (end-diastolic volume on Echo or atrial pressure on PAC are often used as surrogates)
- afterload = maximal tension developed in the ventricular wall during contraction (end-systolic wall stress on Echo), used as a measure of the impedence against which the ventricle must contract.
CARDIAC MUSCLE
- intercalated discs -> transmission of electrical depolarisation easy and produces a coordinated contraction.
- branching cells
- single nucleus
- visible striations
- T-tubules are located on Z-lines (not A-I function)
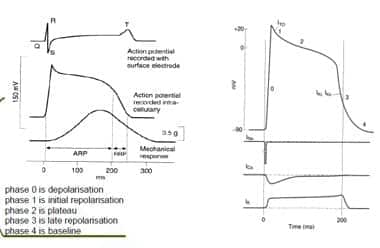
ELECTRICAL PROPERTIES
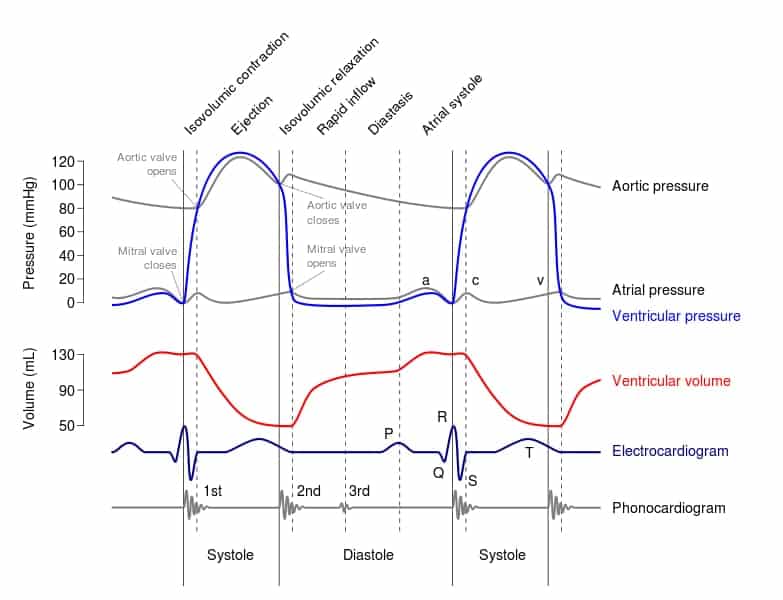
CARDIAC CYCLE
Wigger’s diagram demonstrating the components of the cardiac cycle
- See here for a Flash animation
FRANK-STARLING LAW
- The Frank–Starling law of the heart states that the stroke volume of the heart increases in response to an increase in the volume of blood filling the heart (the end diastolic volume) when all other factors remain constant.
- This reflects the length-tension relationship in myocardial sarcomeres – when stretched by higher volumes, there is more optimal overlap between actin adn myosin in the sarcomeres, resulting in greater contractility.
- At high EDV the sarcomeres become over-stretched, and increased contractility is not possible – this SV fails to increase above a threshold EDV.
HAEMODYNAMICS
Mean arterial pressure (MAP): MAP = Q x SVR
- Q = SV x HR
- SV = Preload, Afterload, Contractility
- SVR = change in P/Q
- Q = DPr4/8nl
Where:
- Q = rate of blood flow
- DP = pressure difference
- r = radius
- l = length
- n = viscosity of blood
Preload determined by:
- venous return
- heart rate
- atrial contraction
- atrial and ventricular pressures during diastole
- compliance of ventricles
Afterload determined by:
- systemic vascular resistance
- aortic compliance
- wall thickness
- chamber radius
- ventricular size
- ventricular volume
Contractility determined by:
- substrate supply
- integrity of myofilaments
- co-ordinated depolarisation
- metabolic/electrolyte homeostasis
- functional muscle mass
- coronary blood flow (hypoxia)
- autonomic tone
- hormones
BLOOD SUPPLY TO HEART
Left Coronary artery
- arises from aortic sinus
- passes behind then to left of pulmonary trunk -> left part of the atrioventricular groove -> lateral round the left border of heart to reach the inferior interventricular groove.
- divides into:
(1) LAD -> diagonal branches
- anterolateral wall of left ventricle
- interventricular septum
- anterior wall of right ventricle
- ventricular apex
(2) Circumflex -> obtuse marginal branches
- left atrium
- posterior wall of left ventricle
- lateral wall of left ventricle
Right Coronary
- from anterior sinus and passes forwards between the pulmonary trunk and right atrium
- it descents in the right part o the atrioventricular groove to anastamose near the apex with corresponding branch of the left coronary artery
- supplies:
- -> lateral wall of the right ventricle
- -> posterior wall of the right ventricle
- -> inferior wall of the left ventricle
- -> sinoatrial node in 55% of patients (via nodal branch)
Dominance
- dominance is determined by which coronary artery supplies the posterior descending artery (PDA)
- in 85% of patients this is supplied by the RCA
Venous Drainage
- 2/3 by coronary sinus
- 1/3 drain directly into cardiac cavity
- vessels draining into the coronary sinus:
- -> great cardiac vein (anterior interventricular groove)
- -> middle cardiac vein (inferior interventricular groove)
- -> small cardiac vein (lower border of heart)
- -> oblique vein (descends obliquely on the back of LA)
- anterior cardiac vein lies in the anterior atrioventricular groove (it drains much of the anterior surface of the heart and opens directly into the RA)
Anatomy Zone — Arterial blood supply to the heart
Anatomy zone — Veins of the heart
References and Links
Social media and web resources
- Cardiovascular physiology concepts by Richard E. Klabunde

Critical Care
Compendium
Chris is an Intensivist and ECMO specialist at The Alfred ICU, where he is Deputy Director (Education). He is a Clinical Adjunct Associate Professor at Monash University, the Lead for the Clinician Educator Incubator programme, and a CICM First Part Examiner.
He is an internationally recognised Clinician Educator with a passion for helping clinicians learn and for improving the clinical performance of individuals and collectives. He was one of the founders of the FOAM movement (Free Open-Access Medical education) has been recognised for his contributions to education with awards from ANZICS, ANZAHPE, and ACEM.
His one great achievement is being the father of three amazing children.
On Bluesky, he is @precordialthump.bsky.social and on the site that Elon has screwed up, he is @precordialthump.
| INTENSIVE | RAGE | Resuscitology | SMACC