Construction workers cough or surfers surprise?
aka Tropical Travel Trouble 013
Guest Post: Dr An Du Thinh (DTM&H)
It can be common to get the ‘twofor’, you know, upon entering the cubicle instead of one patient you get two. Either you know before or get ambushed once you’ve finished the first patient… ‘hey doc, just a quick question… I’m a triple transplant recipient and I wondered if you could run through my anti rejection drug dosing and levels’.
Well today is your lucky day, you get a ‘threefor’, three strapping gents in their mid thirties. No prior medical history and have just returned from the Northern Territory after doing a bit of FIFO work (fly in, fly out). They state they were not installing air-conditioning units (you can scratch through Legionnaires, you keeners) as nothing had been built yet but helping lay the foundations and largely excavating. They had embarked on this mission to earn a quick buck to help fund a surf trip to Thailand and admit to a few practise breaks while off work.
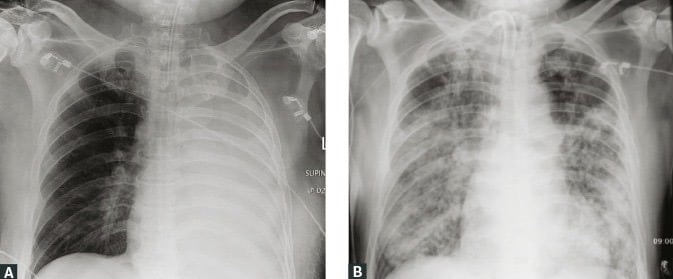
Patient A is complaining of shortness of breath, productive yellow sputum, pleuritic pain on the left. Sats 88%, HR120 sinus, RR 22, BP 116/76, Temp 38.3 and GCS 15.
Patient B feels like crap. He looks like crap as well. Seems a bit confused, short of breath and his buddies had to help drag him in and have put him on the bed. Sats 88%, HR130 sinus, BP 80/58, RR26, Temp 38.8.
X-rays of the two patients are below:
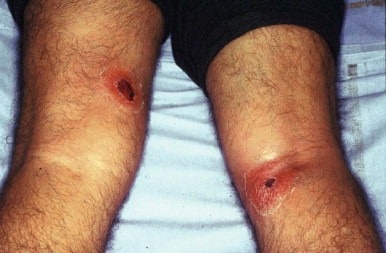
Patient C is okay, he doesn’t have any shortness of breath or chest pain but he does have two wounds on his inside thigh and behind his knee that have been there for the last 2 weeks with some discharge.
All three patients have the same disease. Patient A clearly has pneumonia, patient B is septic with his widespread pneumonia and patient C has chronic wounds/abscesses. What is this disease?
Answer
Melioidosis, it is caused by the Gram-negative rod bacterium Burkholderia pseudomallei.
Together with C.S. Krishnaswami, Whitmore identified the bacteria now known as Burkholderia pseudomallei to be the cause of melioidosis (also known as “Whitmore’s disease”) in opium addicts in Rangoon in 1911. He differentiated it from Burkholderia mallei, the causative agent of glanders, which infects horses and mules (vanishingly rare in humans). Glanders causes nodular lesions in the lungs and ulceration of the mucous membranes in the upper respiratory tract. The acute form results in coughing, fever, and the release of an infectious nasal discharge, followed by septicaemia and death within days for the animals.
…disease somewhat resembling but really easily distinguishable from glanders prevalent among the ill-nourished, neglected, wastrels of the town. In April 1911, we had occasion to perform a post mortem upon the body of a Burman aged 40 years: he had been admitted to hospital for fever of seven days duration and died after three days stay in hospital. During these three days his temperature have been high, ranging from 103 to 104F. His thighs bore numerous marks of morphia injections and in connection with these injections were several subcutaneous abscesses. At the post mortem examination the principal lesion discovered was a peculiar cheesy consolidation of the lungs. The distribution and appearance of this consolidation were those of neither ordinary lobar pneumonia nor tubercular infection, and upon examining smears from the diseased patches a large number of non-Gram-staining bacilli of the size and shape of B. mallei..
Whitmore 1911
Where would you expect to find this disease and where in the environment is it located?
Answer
It is predominantly found in South and East Asia, Northern Australia classically. However, it is generally under-reported due to lack of clinical awareness and diagnostic challenges so realistically it can be found throughout the tropics.
B. pseudomallei is an environmental pathogen and can be found in contaminated soil, water, and plants; transmission from infected animals has also been reported. Long incubation periods have been reported, sometimes up to 62 years. It caused a particular problem in the Vietnam war veterans, particularly for returning winch operators who were exposed to bioaerosols thrown up by the helicopter wash.
How do you get this infection?
Answer
Infection is acquired either by inoculation or inhalation of contaminated soil or water. Most infection is asymptomatic, organisms may remain latent within macrophages and can causes disease many years after infection. Localised abscesses may develop at the site of inoculation which can lead to bacteraemia and dissemination of the organism.
Up to 70% of patients have a predisposing condition. What conditions and risk factors are important?
Answer
- Major
- Diabetes Mellitus
- Alcohol excess leading to liver disease
- Travel to endemic areas with exposure to soil and water
- Minor
- Renal failure and kidney stones
- Immunosuppression with steroids
- Malignancy
- Thalassaemia major
There is no association with HIV itself although if on immunosuppression that is a risk factor.
What are the myriad of clinical presentations and features?
Answer
Many individuals have positive serology without having had obvious clinical symptoms. Acute presentations can be localised or septicaemic disease. The most common form of localised disease is pneumonia, but abscesses may also be found in the skin and soft tissue or organs such as the spleen and liver. Localised disease may lead to subsequent bacteraemia. Post septicaemia, dissemination can occur and cause abscesses in a number of different sites.
A clinic-pathological classification of melioidosis that combines disease localisation, and culture positivity has recently come out of a national melioidosis registry in Sri Lanka (Corea et al). All cases need to be culture-confirmed:
- Blood culture positive or not
- Single or multi-organ system focus or no defined focus
In practice this gives us five presentations: BC pos/no recognised focus; BC pos/unifocal; BC pos/multifocal; BC neg/unifocal; BC neg/multifocal
- Asymptomatic seroconversion – most common as suggested by seroprevalence studies
- Acute infections 85% and symptoms less than 2 months.
- 50% are usually bacteraemic on presentation with 20% in septic shock.
- 50% pulmonary infection (20% in paediatrics)
- 13% cutaneous infection (60% paediatrics) typically at the site of inoculation
- Parenchymal visceral abscesses including spleen, liver, kidneys and prostate (18% or males).
- Intracerebral abcesses and encephalomyelitis.
- 4% bone or joint infection
- Rarely mycotic aneurysms, pericarditis, mediastinal masses, thyroid and scrotal abscesses.
- Chronic infection 11% as defined as a duration >2 months
- Pulmonary infections mimic TB and typically have a upper lobe infiltrate.
- Non-healing cutaneous ulcers unresponsive to antibiotics.
- Reactivation of latent disease (4%). Latent periods of 14-24 years, possibly up to 62 years.
Mean incubation period approximately 9 days (range: 1–21 days), but presents much earlier following inhalational/aspiration events. Below are 3 diagrams to help you visualise the potential presentations and complications.


How is the diagnosis made and why is it under recognised?
Answer
Melioidosis is a neglected tropical disease whose diagnosis in temperate climates is often overlooked until unexpected laboratory results turn up.
None of the clinical features of melioidosis are pathognomonic, and its multiple clinical presentations have a broad differential diagnosis.
The bacterial cause, B. pseudomallei is under recognised from non-sterile sites such as superficial wounds or the respiratory tract, where is may be present in relatively low numbers. In some cases, routine bacteriology tests can mistakenly identify B. pseudomallei as a commensal species.
Even in melioidosis endemic regions, the infection can be seasonal so that out-of-season presentation can be unexpected.
There are a number of investigations you can do as part of your septic work up (see below) but the diagnosis of melioidosis relies heavily on culture, most commonly blood.
- Chest XR to confirm pulmonary involvement and monitor progress. Up to 50% of cases have pneumonia
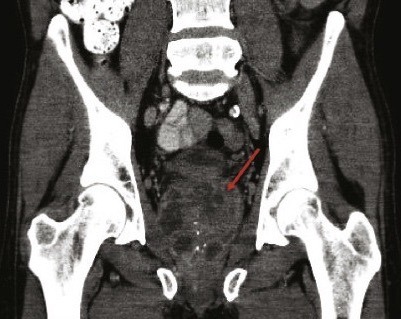
- Ultrasound and/or CT scan to characterise abscesses +/- draining. Dissemination of the bacteria can occur anywhere but typically into the spleen, prostate, liver, kidneys and the parotid gland.
- Septic panel bloods including CRP, which is usually elevated in acute infection.
- Blood culture and cultures from anywhere appropriate including throat, rectal, urine, pus and sputum. Also cultures have a low sensitivity of only 60% and therefore if there is still clinical suspicion, all cultures should be repeated after 3-7 days.
How to diagnose B.pseudomallei and why the lab might miss it:
Firstly, warn the staff, B. pseudomallei is a biosafety level 3 precaution. The US military have it up there with anthrax as a possible biological weapon.
Isolation of bacteria from culture is the standard approach, and B. pseudomallei can grow on most routine media. You might think is great but unfortunately it is often dismissed as a commensal species due to its initial non-specific appearance. The image below shows the classic appearance that the lab hopefully wouldn’t miss:
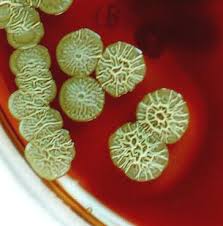
However, initially it can grow as creamy white, smooth rounded colonies and be dismissed as a contaminant or some can be overtly mucoid, similar to Pseudomonas aeruginosa, see below or in this link for clearer examples of this confusion:
To increase your chances the lab should select Ashdown agar or a specific Burkholderia agar. Some bacteria can overgrow B. pseudomallei so to overcome this the lab should add Gentamicin and Colistin which B. pseudomallei is universally resistant to. Plates should be inspected daily and allowed to incubate for at least 4 days.
Molecular techniques are available but are of limited use when applied directly to clinical specimens. PCR assays are used extensively to confirm the identity of suspect bacterial isolates as alternative to MALDI-TOF. Serological tests can detect rising titres of IgG or a raised specific IgM in acute infections, but are far less sensitive than culture in endemic areas and non-specific (false positive) results are known.
What is the management of this disease?
Answer
May start empiric treatment in endemic areas with clinical suspicion. B. pseudomallei is susceptible to the following antibiotics:
- Bacteriocidal beta lactams (activity varies)
- Ceftazadime
- Meropenem
- Imipenem
- Co-amoxiclav
- Bacterio-static
- Doxycycline
- Trimethoprim-sulfamethoxazole
- Most isolate will state susceptibility to piperacillin, ceftriaxone, cefotaxime and chloramphenicol but these agents are clinically less effective and often are not reported in a lot of countries. Beware!
Management:
- Antibiotic therapy:
- Meropenem, first-line therapy in the acutely unwell patient. 25 mg/kg to 1g Q8 hourly. 50 mg/kg to 2g for CNS infections.
- or IV Ceftazidime 50 mg/kg to 2g Q6 hourly.
- TMP-SMX 320/1600 mg Q12 hourly (with folic acid in children 0.1mg/kg up to 5 mg daily), may be added for severe disease.
- 10 – 14 days for isolated pulmonary/cutanous disease/bacteraemia without focus.
- 4 – 8 weeks in severe disease/clinical deterioration/complicated pneumonia, deep seated infection, bone/joint infection, neurological disease.
- Adjunctive therapy:
- Surgical drainage
- Eradication therapy (institute immediately to prevent relapse and continue for 3-6 months guided by response – this is to lower the relapse rate but can still be as high as 25% throughout the patients lifetime)
- TMP-SMX 320/1600mg Q12 hourly.
- Doxycycline can be used if there is TMP-SMX intolerance or adverse drug reactions.
- Amoxicillin/clavulanic acid is reserved for 3rd line due to treatment failure and relapses.
- Post-exposure prophylaxis:
- 21 days of the above oral therapy after discussion with an infectious disease doctor. Usual circumstance would be a high-risk laboratory exposure e.g. penetrating injuries, mouth or eyes or the generation of aerosols outside the safety cabinet.
Fevers persist for a median duration of 9 days, their presence does not indicate failure of treatment.
What can you do to prevent this disease?
Answer
- There is no vaccine but research is ongoing
- Prompt cleaning and sterile dressings for skin abrasions and open wounds
- Avoidance of soil and standing water in endemic areas
- Rubber boots and gloves for agriculture workers, especially in rice-farming cultures (idealistic)
- Drink bottled water and eat cooked food
Case resolutions:
Answer
They all lived happily ever after and went surfing in Thailand, although with the above precautionary measures (Thailand is melioidosis territory, surfing is fine, wading in paddy fields is not).
Patient A was treated with IV meropenem and carried for initially in HDU. He only required 40% FiO2 on optiflow and continued to improve after 4 weeks of IV. His course was interrupted on day 15 by a return of his fevers and a CT showed a pulmonary abscess which was surgically drained. He was placed on 6 months of TMP-SMX.
Patient B became more confused as was intubated after 2L of ringers lactate and a peripheral norepinephrine infusion. Post intubation arterial lines and central lines were placed. He was also placed on meropenem but at a higher dose of 50mg/kg as well as TMP-SMX. Pressor were finally weaned after 10 days and he was discharged after 8 weeks of IV antibiotics. Surprisingly he never developed an abscess. He was also placed on 6 months of TMP-SMX.
Patient C had a quick procedural sedation to I+D his abscesses in emergency which grew B. pseudomallei. He was given a 3 month course of TMP-SMX and his wounds healed without any complications.
Summary:
Answer
Melioidosis is a disease caused from Burkholderia pseudomallei a gram-negative environmental bacterium and accounts for approximately 89,000 deaths per year. Diabetes mellitus is a major risk factor. The tropics including northern Australia are classic endemic areas. The disease ranges from septicaemia to chronic infection. Delay in treatment leads to poor outcome with mortality rates >40%.
The standard diagnosis is from a culture, most often blood. First line treatment is with meropenem or ceftazadime. Patients usually require a prolonged IV course followed by oral eradication therapy with TMP-SMX for 3-6 months. Lifelong risk of relapse is 25% despite treatment.
More resources:
References
- Borton, C. Melioidosis and Glanders, https://patient.info/doctor/melioidosis-and-glanders.htm
- Inglis T. Meliodosis a disease of surprises. LITFL – m.
- Inglis T. Lab without boarders. LITFL
- CDC. Melioidosis, https://www.cdc.gov/melioidosis/index.html
- Chakravorty A and Heath C. Melioidosis: An updated review. AJGP 2019;48(5):1
- Inglis TJJ and Sagripanti J. Environmental Factors That Affect the Survival and Persistence of Burkholderia pseudomallei. Applied and Environmental Microbiology. 2006;72(11):6865-6875
- Koh, G. Melioidosis handout for DTM&H 2018 spring cohort.
- MORU. Melioidosis, melioidosis.info, http://www.melioidosis.info/info.aspx?pageID=104&contentID=1040101
- Nickson, C. Melioidosis, https://litfl.com/melioidosis/
- Sathkumara H et al. Clinical, Bacteriologic, and Geographic Stratification of Melioidosis Emerges from the Sri Lankan National Surveillance Program. AJTM&H 2018;98(2):607-615
- Whitmore A and Krishnaswami CS. A Hitherto Undescribed Infective Disease in Rangoon. The Indian Medical Gazette 1912;47(7):262-267
- Wiersinga WJ et al. Meliodosis. NEJM 2012;367(11):1035-1044
- Wiersinga WJ et al. Meliodosis. Nature Reviews 2018 Article number 17107
Peer Reviewers
• Prof Tim Inglis
CLINICAL CASES
Tropical Travel Trouble
Dr Neil Long BMBS FACEM FRCEM FRCPC. Emergency Physician at Kelowna hospital, British Columbia. Loves the misery of alpine climbing and working in austere environments (namely tertiary trauma centres). Supporter of FOAMed, lifelong education and trying to find that elusive peak performance.



Fascinating post!