Deresuscitation and Positive Fluid Balance
Revised and reviewed 12 July 2015
OVERVIEW
- Deresuscitation specifically refers to ‘Late Goal Directed Fluid Removal (LGFR)’, which involves “aggressive and active fluid removal by means of diuretics and renal replacement therapy with net ultrafiltration” (Malbrain et al, 2014)
- Deresuscitation is also sometimes used to more loosely refer to the the phase of critical illness and/or the care of a critically ill patient, after initial resuscitation and stabilisation. It is characterised by the discontinuation of invasive therapies and a transition to a negative fluid balance.
- Late conservative fluid management (LCFM) is defined as two consecutive days of negative fluid balance within the first week of ICU stay, and is an independent predictor of survival in ICU patients (Malbrain et al, 2014)
POSITIVE FLUID BALANCE
Positive fluid balance is a state of fluid overload resulting from fluid administration during resuscitation and subsequent therapies
- Fluid overload is defined by “a cut off value of 10% of fluid accumulation as this is associated with worse outcomes” (Malbrain et al, 2014)
- Percentage of fluid accumulation can be defined “by dividing the cumulative fluid balance in litre by the patient’s baseline body weight and multiplying by 100%” (Malbrain et al, 2014)
Positive fluid balance is associated with worse morbidity and mortality in multiple studies:
- worse overall mortality in critically ill patients (systematic review by Malbrain et al, 2014)
- increased mortality in patients with acute kidney injury (AKI) (SOAP study)
- prolonged recovery in patients with acute lung injury/ acute respiratory distress syndrome (ALI/ARDS) (FACTT trial); improved mortality in ARDSNET patients if they had negative fluid balance at day 4 (Rosenberg et al, 2009)
- worse mortality in septic shock patients (VAST trial)
- worse morbidity in colorectal surgery patients
- associated with intra-abdominal hypertension (Malbrain et al, 2014)
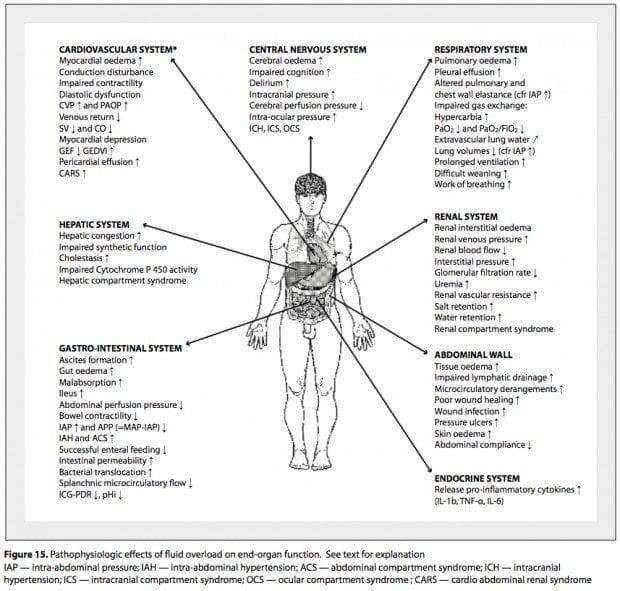
Pathophysiological effects of fluid overload
The kidneys are susceptible to functional impairment due to positive fluid balance
- postive fluid balance causes interstitial edema and intrabdominal hypertension
- these effects impair kidney function and cause oliguria
- thus positive fluid balance counteracts its own resolution
MALBRAIN ET AL’S “ROSE” CONCEPT OF PHASES OF CRITICAL ILLNESS
Resuscitation phase (R)
- Salvage or rescue treatment with fluids administered quickly as a bolus (4 mL kg-1 over 10 to 15 minutes)
- The goal is early adequate goal directed fluid management (EAFM), fluid balance must be positive and the suggested resuscitation targets are:
MAP > 65 mm Hg, CI > 2.5 L min-1m-2, PPV < 12%, LVEDAI > 8 cm m-2.
Optimisation phase (O)
- occurs within hours
- ischaemia and reperfusion
- degree of positive fluid balance may be marker of severity in this phase
- risk of polycompartment syndrome
- unstable, compensated shock state requiring titrating of fluids to cardiac output
- Targets: MAP > 65 mm Hg , CI > 2.5 L min-1m-2, PPV < 14%, LVEDAI 8−12 cm-1m-2, IAP (< 15 mm Hg ) is monitored and APP (> 55 mm Hg ) is calculated. Preload optimised with GEDVI 640—800 mL m-2
Stabilisation phase (S)
- evolves over days
- fluid therapy only for normal maintenance and replacement
- absence of shock or threat of shock
- Monitor daily body weight, fluid balance and organ function
- Targets: neutral or negative fluid balance; EVLWI < 10−12 mL kg-1 PBW, PVPI < 2.5, IAP < 15 mm Hg , APP > 55 mm Hg , COP > 16−18 mm Hg , and CLI < 60
Evacuation phase (E)
- patients who do not transition from the ‘ebb’ phase of shock to the ‘flow’ phase after the ‘2nd hit’ develop global increased permeability syndrome (GIPS)
- fluid overload casues end-organ dysfunction
- requires late goal directed fluid removal (“de-resuscitation”) to achieve negative fluid balance
- need to avoid over enthusiastic fluid removal resulting in hypovolaemia
DERESUSCITATION
Recommendations
- “a goal of a zero to negative fluid balance by day 3 and to keep the cumulative fluid balance on day 7 as low as possible (Grade 2B)” (Malbrain et al, 2014)
- “diuretics or renal replacement therapy (in combination with albumin) can be used to mobilise fluids in haemodynamically stable patients with intra-abdominal hypertension and a positive cumulative fluid balance after the acute resuscitation has been completed and the inciting issues/source control have been addressed (Grade 2D)” (Malbrain et al, 2014)
Cordeman et al (2012) suggest the ‘PAL’ approach
- high PEEP for 30 minutes (at least equal to intra-abdominal pressure) – to drive fluid from the alveoli into the interstitium, then
- Albumin administration (e.g. 2 x 100 mL 20% albumin over 60 minutes on Day 1, then titrated to albumin >30 g/L) – to pull fluid from the interstitium into the circulation
- Frusemide (‘Lasix’) infusion started 60 minutes after albumin at 60 mg/h for 4 hors, then titrated between5 and 20 mg/h to maintain >100 mL/h urine output
- Use of this approach in patients with “acute lung injury” as part of a pilot study was associated with a negative fluid balance, a reduction of EVLWI and IAP, and decreased duration of mechanical ventilation without compromising organ function.
References and Links
Journal articles
- Bouchard J, Mehta RL. Fluid balance issues in the critically ill patient. Contrib Nephrol. 2010;164:69-78. [pubmed]
- Cordemans C, De Laet I, Van Regenmortel N, et al. Aiming for a negative fluid balance in patients with acute lung injury and increased intra-abdominal pressure: a pilot study looking at the effects of PAL-treatment. Ann Intensive Care. 2012;2 Suppl 1:S15. [pubmed] [free full text]
- Gonzalez F, Vincent F. The fluid balance in the critically ill patients: what are we talking about? Minerva Anestesiol. 2011;77:(8)766-7. [pubmed]
- Malbrain ML, Marik PE, Witters I, Cordemans C, Kirkpatrick AW, Roberts DJ, Van Regenmortel N. Fluid overload, de-resuscitation, and outcomes in critically ill or injured patients: a systematic review with suggestions for clinical practice. Anaesthesiol Intensive Ther. 2014 Nov-Dec;46(5):361-80. doi: 10.5603/AIT.2014.0060. PMID: 25432556. [free full text]
- Rosenberg AL, Dechert RE, Park PK, Bartlett RH, et al. Review of a large clinical series: association of cumulative fluid balance on outcome in acute lung injury: a retrospective review of the ARDSnet tidal volume study cohort. Journal of intensive care medicine. 2009; 24(1):35-46. [pubmed]

Critical Care
Compendium
Chris is an Intensivist and ECMO specialist at The Alfred ICU, where he is Deputy Director (Education). He is a Clinical Adjunct Associate Professor at Monash University, the Lead for the Clinician Educator Incubator programme, and a CICM First Part Examiner.
He is an internationally recognised Clinician Educator with a passion for helping clinicians learn and for improving the clinical performance of individuals and collectives. He was one of the founders of the FOAM movement (Free Open-Access Medical education) has been recognised for his contributions to education with awards from ANZICS, ANZAHPE, and ACEM.
His one great achievement is being the father of three amazing children.
On Bluesky, he is @precordialthump.bsky.social and on the site that Elon has screwed up, he is @precordialthump.
| INTENSIVE | RAGE | Resuscitology | SMACC