ECMO Literature Summaries
Peek GJ, et al; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009 Oct 17;374(9698):1351-63.PMID: 19762075.
- UK
- MC-RCT
- n = 180
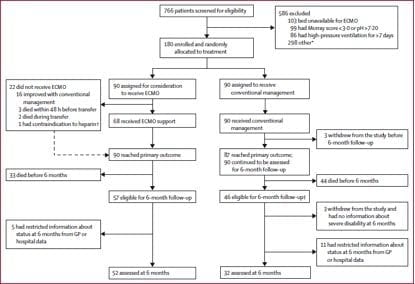
- randomised to consideration for ECMO (90) -> 68 received ECMO vs conventional treatment (90)
- inclusion criteria: adults, reversible respiratory failure, Murray score of >3.0 (PaO2/FiO2 ratio, PEEP, compliance, CXR appearance, FiO2 1.0), pH 0.8, ventilation > 7 days, contraindications to anticoagulation, contraindication to continuation to active treatment.
- primary outcome = death or severe disability @ 6 months
- secondary outcomes = duration of ventilation, use of HFOV or jet ventilation, use of NO, prone positioning, steroids, duration of ICU stay, duration of hospital stay, method of ECMO, duration of ECMO, blood flow, sweep flow.
- intention to treat analysis
- data on resource use and economic outcomes (quality-adjusted life years) collected
- ECMO patients -> Leicester
- conventional ventilation patients -> tertial intensive care units
- all inward ECMO patients done by ECMO team -> but did not provide ECMO
- 85 patient arrived in Glenfield (5 died pre or during transfer) -> 17 treated with gentle ventilation of who 14 survived (82%)
- 68 treated with ECMO of whom 42 survived (63%)
- only 68 of the 90 (75%) of the patients considered for ECMO received it
- survival in the non-ECMO consideration group was 47%.
- important differences between the groups:
(1) almost a quarter of the consideration for ECMO group did not receive ECMO
(2) MARS = molecular albumin recirculating system offered at Leicester to those transferred
(3) steroids used at Leicester in consideration for ECMO group
(4) ARDS net ventilation used more in patients transferred to Leicester
- 12 hour window in Leicester to optimize patient -> if failed -> ECMO
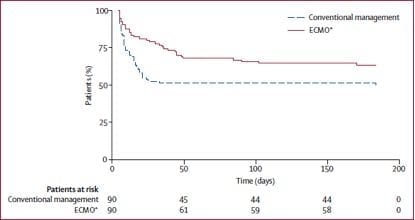
-> concluded that ECMO produced increased survival and minimises severe disability, and is cost effective
-> relative risk of death or severe disability @ 6 month in ECMO vs control = 0.69 (NNT = 5)
-> strengths: pragmatic
-> weakness: should have only enrolled patients for ECMO once arrived in Leicester!
Australia and New Zealand Extracorporeal Membrane Oxygenation (ANZ ECMO) Influenza Investigators, Davies A, et al. Extracorporeal Membrane Oxygenation for 2009 Influenza A(H1N1) Acute Respiratory Distress Syndrome. JAMA. 2009 Nov 4;302(17):1888-95. PMID: 19822628.
- observational study
- Australasian
- June – August 2009
- n = 201 (requiring mechanical ventilation)
- 30% received (68)
- mortality rate in ECMO group = 21% (lower than previous documented)
- mortality in non-ECMO group = 13%
- Conclusion: Uee of ECMO associated with low rates of mortality in severe hypoxia in H1N1
Mitchell MD, Mikkelsen ME, Umscheid CA, Lee I, Fuchs BD, Halpern SD. A systematic review to inform institutional decisions about the use of extracorporeal membrane oxygenation during the H1N1 influenza pandemic. Crit Care Med. 2010 Jun;38(6):1398-404. PMID: 20400902.
- Conclusion: insufficient evidence for ECMO use among patients with H1N1.

Critical Care
Compendium
Chris is an Intensivist and ECMO specialist at The Alfred ICU, where he is Deputy Director (Education). He is a Clinical Adjunct Associate Professor at Monash University, the Lead for the Clinician Educator Incubator programme, and a CICM First Part Examiner.
He is an internationally recognised Clinician Educator with a passion for helping clinicians learn and for improving the clinical performance of individuals and collectives. He was one of the founders of the FOAM movement (Free Open-Access Medical education) has been recognised for his contributions to education with awards from ANZICS, ANZAHPE, and ACEM.
His one great achievement is being the father of three amazing children.
On Bluesky, he is @precordialthump.bsky.social and on the site that Elon has screwed up, he is @precordialthump.
| INTENSIVE | RAGE | Resuscitology | SMACC