PiCCO
OVERVIEW
- PiCCO is a cardiac output monitor that combines pulse contour analysis and transpulmonary thermodilution technique
- PiCCO2 includes continuous ScvO2 monitoring (CeVOX probe via standard CVC)
USE
Haemodynamic monitoring
- especially if complex mixed forms of shock (e.g. septic and cardiogenic)
- PAC unavailable or contra-indicated
Contra-indicated when factors that cause inaccurate measurements are present:
- intracardiac shunts
- aortic aneurysm
- aortic stenosis
- pneumonectomy
- pulmonary embolus
- in the presence of a balloon pump and in unstable arrhythmias
DESCRIPTION
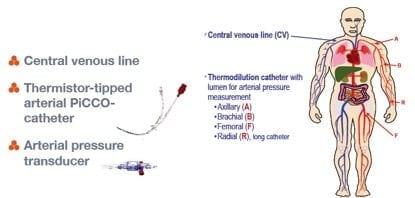
- PiCCO catheter is arterial line with a thermistor on the end
- provides complete haemodynamic monitoring by combining pulse contour analysis to provide a continuous display of cardiac output using a modified version of Wesseling algorithm combined with a transpulmonary thermodilution technique
- pressure transducer
- PiCCO monitor:
METHOD OF INSERTION/ USE
- insert a central line
- place PiCCO into a large artery, usually the femoral (axillary is an alternative)
- attach PiCCO via a pressure transducer to the PiCCO monitor
- enter patient details prior to calibration
- administer a known volume of cold normal saline via central line injection port
- PiCCO detects temperature difference and generates a dissipation curve to which the Stewart Hamilton equation is applied to calculate CO.
- Other measures generated include:
— preload: global end-diastolic blood volume (GEDV) and intrathoracic blood volume (ITBV)
— extravascular lung water (EVLW) which is a sensitive indicator of pulmonary oedema.
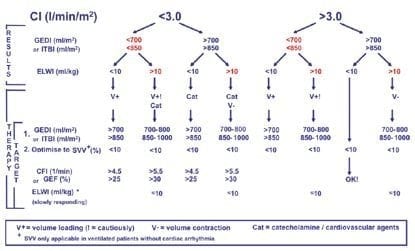
— arterial BP, HR, stroke volume (SV), systemic vascular resistance (SVR), and cardiac function index (CI). - decision trees is provided with PiCCO to help interpret these parameters in terms of haemodynamic status
Thermodilution
- calibration technique
- cold or room temperature saline bolus -> measured by arterial thermistor
- temperature change vs time plotted
- parameters derived:
Cardiac output (Q) and cardiac index (CI)
Global end-diastolic volume (GEDV) – all four chambers
Global ejection fraction – ratio of 4 stroke volumes divided by GEDV (can detect ventricular dysfunction)
Intrathoracic blood volume (ITBV) – volume in heart + pulmonary vessels
Extravascular lung water (EVLW) – water content in lungs
Arterial pulse contour analysis
- ongoing measurement -> providing continuous beat-by-beat parameters obtained from shape of arterial pressure wave.
- parameters derived:
Pulse continuous cardiac output (PCCO)
SVR
Stroke volume variation (SVV) – sensitivity of heart to cyclic changes in preload induced by respiration.
dPmax – slope of pressure vs time trace = closely approximates contractility of LV
Central Venous Oximetry
- measures ScvO2 by spectrophotometry
- displayed continuously
COMPLICATIONS
- usual arterial line complications
- cold fluid in haemodynamically unstable patients – volume and temperature effects
- effects of acting on incorrect measurements/ interpretations
OTHER INFORMATION
Summary of useful values and normal ranges
Q = SV x HR
SV – preload, afterload and contractility
Preload
- Global end-diastolic index (GEDI) – 680-800mL/m2
- Intra-thoracic blood index (ITBI) – 850-1000mL/m2
- Stroke volume variation (SVV) – 3.0 L/min/m2
Afterload
- Systemic vascular resistance index (SVRI) – 1700-2400 dyn.sec.cm-5.m2
Heart function/Contractility
- Cardiac index (CI)/Pulse contour cardiac index (PCCI) – 3.0-5.0L/min/m2
- Global ejection fraction (GEF) – 25-35%
Lung Function
- Extra vascular lung water index (ELWI) – 3-7mL/kg
Oxygen Delivery
- ScvO2 – 70-80%
Summary of Haemodynamic Goals
- CI > 3.0 L/min/m2
- appropriate preload: GEDI of 700-800mL/m2 or ITBI of 850-1000mL/m2 or SVV < 10%
- minimal extra vascular lung water: ELWI 3-7mL/kg
- ScvO2 70-80%
PROS AND CONS
Advantages of PiCCO
- transpulmonary thermodilution (vs only right sided thermodilution)
- less invasive
- dynamic continuous measurement
- also measures extra-vascular lung water
- can stay in patient for up to 10 days (unlike PAC – 72 hours)
- no CXR required
- claimed to be cheaper
- patients usually already require a CVL and arterial line
- ITBV and GEDV are better indicators or preload than CVP and PCWP and not influenced by mechanical ventilation
- EVLW shown to have clear correlation in severity of ARDS, number of ventilator days, ICU duration and mortality
Disadvantages of PiCCO
- cannot be used with an intra-aortic balloon pump
- should be recalibrated with changes in position, therapy or condition to account for compliance of vascular bed
- in obese patients, EVLW underestimated as related to weight of patient (use IBW)
- EVLW only measured in parts of the lung that are perfused (underestimated post pneumonectomy)
- AAA raises GEDV and ITBB
- severe AR may result in an inaccurate thermodilution wash out curve
EVIDENCE
PiCCO vs Physician – Critical Care 2007 11 (suppl 2), page 283
- EVLW and GEDV vs clinical exam, CVP and CXR
-> leg oedema and increased CVP does not exclude underfilling and is a poor predictor of EVLWI
-> EVLW associated with audible crepitations
-> EVLW underestimated by CXR
PiCCO vs PAC – Uchino and Bellomo – Critical Care 2006, 10: R174
- MRCT
- 4 countries
- 342 catheters (PiCCO 192, PAC 150)
-> PiCCO associated with greater positive fluid balance and fewer ventilator-free days
-> no major influence on outcomes
-> positive fluid balance significant predictor of hospital mortality (independent of type of monitoring used)
References and Links
FOAM and web resources
- Deranged Physiology — PiCCO

Critical Care
Compendium
Chris is an Intensivist and ECMO specialist at The Alfred ICU, where he is Deputy Director (Education). He is a Clinical Adjunct Associate Professor at Monash University, the Lead for the Clinician Educator Incubator programme, and a CICM First Part Examiner.
He is an internationally recognised Clinician Educator with a passion for helping clinicians learn and for improving the clinical performance of individuals and collectives. He was one of the founders of the FOAM movement (Free Open-Access Medical education) has been recognised for his contributions to education with awards from ANZICS, ANZAHPE, and ACEM.
His one great achievement is being the father of three amazing children.
On Bluesky, he is @precordialthump.bsky.social and on the site that Elon has screwed up, he is @precordialthump.
| INTENSIVE | RAGE | Resuscitology | SMACC


Nice summary. I stille have one unanswered question, that you hopefully can help me answer: what body weight should be used? Weight at admission IBW, current body weight (including positive fluid balance)?
The choice of weight will affect all indexed values and therapeutic recommendations.