Pulmonary Embolism
OVERVIEW
- Pulmonary embolism (PTE, PE) ranges from asymptomatic to a life threatening catastrophe
- PE occurs when a deep vein thrombosis migrates to the pulmonary arterial tree
Types
- massive PE is defined as acute PE with obstructive shock or SBP <90 mmHg
- submassive PE is acute PE without systemic hypotension (SBP ≥90 mm Hg) but with either RV dysfunction or myocardial necrosis
- those with none of the above severe features are non-massive or low risk PEs
PATHOPHYSIOLOGY
- effects are proportional to the rapidity and degree of obstruction
- increased PVR -> RVF -> obstructive shock
- increased alveolar dead space -> V/Q mismatch -> pulmonary vasoconstriction to optimize gas exchange
- pulmonary infarction
- chronic pulmonary hypertension can ensue
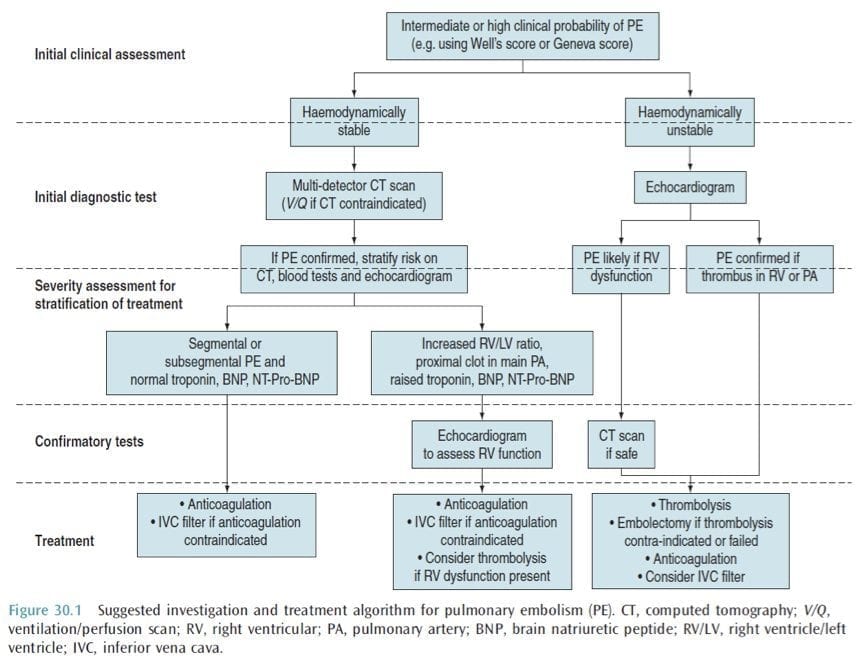
OVERALL APPROACH
CLINICAL FEATURES
History
- may be asymptomatic
- SOB
- pleuritic chest pain
- apprehension
- cough
- haemotypsis
- leg pain
- collapse = massive PE
- acute cardiovascular collapse
Examination
- pale, mottled skin
- tachypnoea
- tachycardia
- signs of DVT
- hypotension
- altered LOC
- elevated JVP
- parasternal heave
- loud P2
- central cyanosis
RISK FACTORS
Major (relative risk 5-20) – SLOMMP
- Surgery – major abdominal/pelvic, hip/knee replacements, post ICU
- Lower limb problems – #, varicose veins
- Obstetrics – late pregnancy, C/S, puerperium
- Malignancy – abdominal/pelvic, advanced/metastatic
- Mobility – hospitalization, institutional care
- Previous VTE
Minor (relative risk 2-4) – COM
- Cardiovascular – congenital heart disease, CHF, HT, superficial venous thrombosis, CVL
- Oestrogens – OCP, HRT
- Miscellaneous – COPD, neurological disability, occult malignancy, thrombotic disorder, long distance travel, obesity, other (IBD, nephrotic syndrome, dialysis, myeloproliferative disorders, paroxysmal nocturnal haemoglobinuria, Bechet’s disease)
Thrombophillias
- Factor V Leiden mutation
- Prothombin gene mutation
- Hyperhomocysteinaemia
- Antiphospholipid antibody syndrome
- Deficiency of antithrombin III, protein C or protein S
- High concentrations of factor VIII or XI
- Increased lipoprotein (a) -> test in those < 50years with recurrent or a strong FHx
INVESTIGATIONS
- goal = confirm diagnosis + assess severity
- normal compliance and peak pressures on ventilator
- increased PACO2 -> ETCO2 to gradient
- hypoxaemia
ECG:
-> mostly normal -> sinus tachycardia -> SI, QIII, TIII -> non specific ST changes or TWI in anterior leads (right heart strain), -> right axis deviation -> s wave (I and aVL) > 1.5mm -> Q wave in III and aVF -> p pulmonale -> RBBB
- CXR: rules out other pathology, focal oligaemia, wedge density (pulmonary infarction)
- ABG: reduced PaO2 in keeping with size of PE, metabolic acidosis with circulatory collapse, respiratory alkalosis
- D-Dimer: reassuring if negative to exclude PE, use in conjunction with clinical probability
- TNT: elevation is associated with adverse outcome even in normotensive patients, also associated with haemodynamic instability in patients with non-massive PE
- BNP and NT-terminal BNP: if low correlates well with uneventful course
- CTPA: as good as pulmonary angiography (gold standard), can calculate RV/LV ratio (>0.9) = severe
- ECHO: RV dialation, paradoxical septal motion towards the LV, TR, RVF, PHT or PA thrombus on TOE,
- US: leg veins (not as accurate as initially thought -> have low threshold to re-scan)
- V/Q scan: only really used now when CT is contraindicated (normal scan, low, intermediate and high probability with various criteria)
- MRI – high rate of technical difficulty and insufficient sensitivity (PIOPED III study)
MANAGEMENT
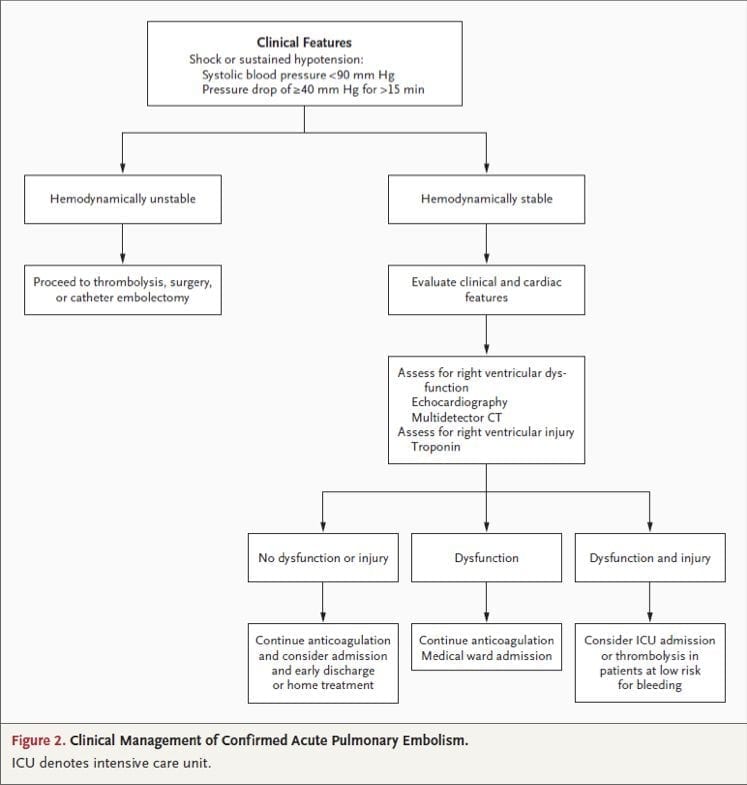
Grade severity of PE
- MASSIVE – haemodynamically unstable -> thrombolyse/embolectomy
- SUB-MASSIVE – haemodynamically stable with evidence of RV dysfunction -> strongly consider thrombolysis/embolectomy but need to balance risk of bleeding
- NON-MASSIVE – haemodynamically stable with normal RV function -> anticoagulation
Management Goals
(1) prevent further embolism (2) removal of emboli (massive or sub-massive) (3) haemodynamic support (massive)
Resuscitation
- A – may need intubation if in cardiovascular collapse or cardiac arrest
- B – high flow O2 as a pulmonary vasodilator, ventilation to optimize V/Q mismatch, hyperventilation to clear CO2
- C – invasive monitoring, fluid management to optimize right ventricular function, inotropic support, cautious fluid boluses, use milrinone, noradrenaline or adrenaline (rather than alpha agonists)
Specific Treatment
THROMBOLYSIS
- not used in non-massive or low risk PEs
- use in submassive PEs is contentious – doesn’t reduce mortality but does reduce deterioration
- can be used up to 14 days after symptoms begin
- PE resolve more quickly than with heparin alone
- as successful as embolectomy in massive PE (earlier the better)
- indicated in patients with RV compromise + haemodynamically unstable
- rTPA 1.5mg/kg is maximum dose (as good through peripheral IV or CVL)
- alteplase 100 mg (0.6mg/kg) as a continuous infusion over 2 hours
- contraindications: absolute – bleeding, recent stroke, HI, current GI bleeding, relative – PUD, surgery within 7 day, prolonged CPR
- follow straight away with heparin
- if bleeds -> FFP and anti-fibrinolytics
ANTICOAGULATION
- use UFH as first dose, in massive PE and where reversal may be required
- start immediately when there is a suspicion (prior to imaging)
- LMWH as good as heparin
- give straight after thrombolysis
- then needs warfarin (INR 2-3)
SURGICAL
- embolectomy (massive PE and unresponsive to thrombolysis or is contraindicated)
- right heart catheterisation with clot destruction
- IVC filter (high risk of further embolic or recurrent PE despite adequate anticoagulation, contraindications to anticoagulation, extensive DVT, massive PE)
OTHER THERAPIES
- ECMO – its role in PE is not established but anecdotally is very effective
- IABP – has been used with vasoactive medications
- Nitric oxide – has been used with some effect
UNDERLYING CAUSE
- prophylaxis
- modify risk factors
- diagnose thrombophillia
METABOLIC
- proportional to the degree of shock
- may have a metabolic acidosis from hyperlactataemia from circulatory compromise
References and Links
LITFL
- Burns E. ECG changes in Pulmonary Embolism ECG library
- Cadogan M. CXR eponyms in pulmonary embolism. LITFL
Journal articles
- Agnelli G, Becattini C. Acute pulmonary embolism. N Engl J Med. 2010 Jul 15;363(3):266-74.
- Jaff MR et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011 Apr 26;123(16):1788-830.

Critical Care
Compendium
Chris is an Intensivist and ECMO specialist at The Alfred ICU, where he is Deputy Director (Education). He is a Clinical Adjunct Associate Professor at Monash University, the Lead for the Clinician Educator Incubator programme, and a CICM First Part Examiner.
He is an internationally recognised Clinician Educator with a passion for helping clinicians learn and for improving the clinical performance of individuals and collectives. He was one of the founders of the FOAM movement (Free Open-Access Medical education) has been recognised for his contributions to education with awards from ANZICS, ANZAHPE, and ACEM.
His one great achievement is being the father of three amazing children.
On Bluesky, he is @precordialthump.bsky.social and on the site that Elon has screwed up, he is @precordialthump.
| INTENSIVE | RAGE | Resuscitology | SMACC
