Selective Lung Ventilation
Reviewed and revised 3 September 2015
OVERVIEW
- Selective Lung Ventilation involves the isolation of one lung from the other allowing independent ventilation.
- Usually performed with a double lumen tube (DLT); alternatives are the use of a univent tube or an endobronchial blocker
RATIONALE
Anatomical lung separation
- Massive hemoptysis (protect normal lung from blood)
- Whole lung lavage for pulmonary alveolar proteinosis (lavage worst lung first)
- Copious secretions (e.g. bronchiectasis, lung abscess) (protect normal lung from pus)
Physiological lung separation
- Unilateral parenchymal injury:
— Aspiration
— Pulmonary contusion
— Pneumonia
— Unilateral pulmonary edema - Single lung transplant (post operative complications)
— e.g. COPD patient with lung transplant requiring different ventilation strategies for each lung - Bronchopleural fistula
— allow ventilation of normal lung without worsening leak through fistula - Unilateral bronchospasm
- Thoracic surgery e.g. pneumonectomy
— right-sided DLT only used if left pneumonectomy or surgery on left main bronchus
DOUBLE LUMEN TUBES
- right or left depending on bronchus they are designed to intubate
- right sided tubes have a slit to be positioned to facilitate ventilation of RUL
- size = external diameter of tube (French Gauge – 26-41 Fr)
- males; 39-41 Fr
- females; 37-39 Fr
- children use bronchial blocker technique
- remember in an emergency (ie. pulmonary haemorrhage) a standard ETT can be advanced into non-diseased lung (a bougie may help facilitate this)
Table below give diameter of bronchus (usually left) measured on PA CXR (magnifies air bronchogram by 10%)
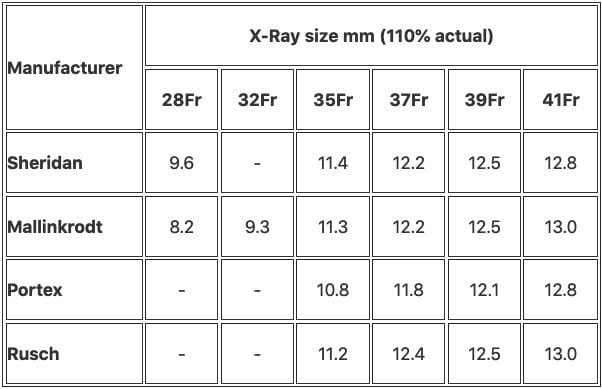
- bronchoscope must be a narrow scope (<4 mm diameter)
- Mallinckrodt; high volume, low pressure cuff, bronchial tube + pilot balloon blue, radioopaque marker stripe running to tip of bronchial lumen
INSERTION
Technique
- ensure lumens are patent and individual cuffs are working
- anaesthetise and paralyse patient
- direct laryngoscopy
- insertion of endotracheal tube with bronchial concave curve facing anteriorly
- as tip passes through larynx rotate the ETT anti-clockwise 90 degrees until resistance met
- once trachea cannulated and tracheal cuff just below the vocal cords, inflate tracheal cuff and ensure ventilation of both lungs via inspection and auscultation
- check ventilation through bronchial lumen (clamp off gas flow to tracheal lumen at Y connector and open the tracheal sealing cap to air) – inflate bronchial cuff 1mL at a time until leak stops
- check whether can isolate other lung via tracheal lumen – close the sealing cap, remove Y connector and ventilate
Trouble shooting techniques include
- use stylet to help place
- using a bougie as an introducer
- using fiberoptic bronchoscope to cannulate bronchus and rail road tube over scope
- clinically check ventilation once changes have been made between normal ventilation and isolated lung ventilation.
- once tracheal cuff below cords, rotate tube towards bronchus that is to be cannulated, turn patient’s head to opposite side, gently slide tube down until resistance felt
INITIATING
- start with typical ventilation settings (FiO2 0.3, TV 6-8 mL/kg)
- if PAW >35cmH2O think: mechanical problem, malposition, obstruction
adjust ventilator setting to keep PAW <30cmH2O
WEANING TO TWO LUNG VENTILATION
- gently suction non-ventilated lung
- close sealing cap on lumen to non-ventilated lung and remove clamp on Y connector
- manually ventilate to re-expand collapsed lung (may need pressures up to 35-40cmH2O)
- return to two lung mechanical ventilation
PROBLEMS
Problems with DLT /SLV
- creates shunt (blood flow to non-ventilated / collapsed lung) resulting in hypoxaemia
- sizing and placement of DLT can be difficult
- DLT can be easily malpositioned during patient movement or care (e.g. in ICU)
- muscosal ischemia or trauma and tracheo-bronchial stenosis from cuff inflation and repositioning
- risk of bronchial rupture
- expertise required for insertion
- need for skilled and intensive nursing and specialised monitoring
- need ready availability of fibreoptic bronchoscopy
MANAGEMENT OF HYPOXIA DURING SLV
Objectives
- minimise atelectasis in the ventilated lung
- minimise shunt in the non-ventilated lung
Approach
- Set initial tidal volume at 10 ml/kg and adjust RR to maintain normocapnia
- Use FIO2 of 0.5 initially and increase to 1.0 if required
- Ensure proper tube position (auscultate, bronchoscopy)
- suction regularly
- Apply CPAP to the non-ventilated lung to expand it just enough so as not to interfere with the surgery, thus reduce shunt.
- Application of PEEP to the ventilated lung may reduce atelectasis but oxygenation may deteriorate due to increase in shunt through the other lung.
- Oxygenation can be insufflated into the non-ventilated lung via a suction catheter. Alternatively, the non-ventilated lung can be inflated briefly with 100% oxygen periodically.
- Persistent hypoxia that does not respond to the above manoeuvres may require two-lung ventilation with 100% O2. Failing this, clamping of the pulmonary artery (of the surgical lung) should improve oxygenation.
VIDEO
From Dr Gallagher’s Neighbourhood:
References and Links
CCC Ventilation Series
Modes: Adaptive Support Ventilation (ASV), Airway Pressure Release Ventilation (APRV), High Frequency Oscillation Ventilation (HFOV), High Frequency Ventilation (HFV), Modes of ventilation, Non-Invasive Ventilation (NIV), Spontaneous breathing and mechanical ventilation
Conditions: Acute Respiratory Distress Syndrome (ARDS), ARDS Definitions, ARDS Literature Summaries, Asthma, Bronchopleural Fistula, Burns, Oxygenation and Ventilation, COPD, Haemoptysis, Improving Oxygenation in ARDS, NIV and Asthma, NIV and the Critically Ill, Ventilator Induced Lung Injury (VILI), Volutrauma
Strategies: ARDSnet Ventilation, Open lung approach, Oxygen Saturation Targets, Protective Lung Ventilation, Recruitment manoeuvres in ARDS, Sedation pauses, Selective Lung Ventilation
Adjuncts: Adjunctive Respiratory Therapies, ECMO Overview, Heliox, Neuromuscular blockade in ARDS, Prone positioning and Mechanical Ventilation
Situations: Cuff leak, Difficulty weaning, High Airway Pressures, Post-Intubation Care, Post-intubation hypoxia
Troubleshooting: Autotriggering of the ventilator, High airway and alveolar pressures / pressure alarm, Ventilator Dyssynchrony
Investigation / Indices: A-a gradient, Capnography and waveforms, Electrical Impedance Tomography, Indices that predict difficult weaning, PaO2/FiO2 Ratio (PF), Transpulmonary pressure (TPP)
Extubation: Cuff Leak Test, Extubation Assessment in ED, Extubation Assessment in ICU, NIV for weaning, Post-Extubation Stridor, Spontaneous breathing trial, Unplanned extubation, Weaning from mechanical ventilation
Core Knowledge: Basics of Mechanical Ventilation, Driving Pressure, Dynamic pressure-volume loops, flow versus time graph, flow volume loops, Indications and complications, Intrinsic PEEP (autoPEEP), Oxygen Haemoglobin Dissociation Curve, Positive End Expiratory Pressure (PEEP), Pulmonary Mechanics, Pressure Vs Time Graph, Pressure vs Volume Loop, Setting up a ventilator, Ventilator waveform analysis, Volume vs time graph
Equipment: Capnography and CO2 Detector, Heat and Moisture Exchanger (HME), Ideal helicopter ventilator, Wet Circuit
MISC: Sedation in ICU, Ventilation literature summaries
LITFL
- CCC — Bronchial blocker
Journal articles
- Anantham D, Jagadesan R, Tiew PE. Clinical review: Independent lung ventilation in critical care. Crit Care. 2005;9(6):594-600. PMC1414047.
- Brodsky JB. Lung separation and the difficult airway. Br J Anaesth. 2009 Dec;103 Suppl 1:i66-75. doi: 10.1093/bja/aep262.PMID: 20007992.
- Campos JH, Ueda K. Lung separation in the morbidly obese patient. Anesthesiol Res Pract. 2012;2012:207598. PMC3287015.
- Campos JH. An update on bronchial blockers during lung separation techniques in adults. Anesth Analg. 2003 Nov;97(5):1266-74. Review. Erratum in: Anesth Analg. 2004 Jan;98(1):131. PMID: 14570636. [Free Fulltext]
- Karzai W, Schwarzkopf K. Hypoxemia during one-lung ventilation: prediction, prevention, and treatment. Anesthesiology. 2009 Jun;110(6):1402-11. PMID: 19417615. [Free Fulltext]
FOAM and web resources
- W. John Russel — Insertion of a double lumen tube
Critical Care
Compendium
Chris is an Intensivist and ECMO specialist at The Alfred ICU, where he is Deputy Director (Education). He is a Clinical Adjunct Associate Professor at Monash University, the Lead for the Clinician Educator Incubator programme, and a CICM First Part Examiner.
He is an internationally recognised Clinician Educator with a passion for helping clinicians learn and for improving the clinical performance of individuals and collectives. He was one of the founders of the FOAM movement (Free Open-Access Medical education) has been recognised for his contributions to education with awards from ANZICS, ANZAHPE, and ACEM.
His one great achievement is being the father of three amazing children.
On Bluesky, he is @precordialthump.bsky.social and on the site that Elon has screwed up, he is @precordialthump.
| INTENSIVE | RAGE | Resuscitology | SMACC
